1. Introduction
Rechargeable lithium-ion batteries (LIBs) were first introduced into the battery market by Sony and Asahi in 1991. Their production has seen massive growth led by Japanese companies such as Panasonic and Hitachi and centering on the information technology (IT) device industry.1) Since 2000, Korean companies such as LG Chemical, Samsung SDI, and Chinese manufacturers including ATL and BYD have entered the market. With the development of high capacity battery technologies, lithium ion batteries have become widely used in commercial industries mainly including smart phones, tablet PCs, laptops, and automobiles because of their high energy density and long life-time compared with the other types of rechargeable batteries.2) The motivation for using a lithium-based battery technology rather than that of a primary (non-rechargeable) battery such as alkaline, mercury, or zinc chloride, relied initially on the fact that Li is the most electropositive (−3.04 V versus standard hydrogen electrode; SHE) and lightest metal (equivalent weight M = 6.94 g·mol−1, and specific gravity r = 0.53 g·cm−3). The advantages of Li metal for batteries were first demonstrated in the 1970s with the assembly of primary Li cells.3,4) Owing to their high theoretical capacity and fast Li-ion mobility, Li-ion batteries rapidly found applications as power sources for IT based applications as well as implantable medical devices.
As the global market for lithium-ion secondary batteries is growing and expected to grow to several times the size of the present market in 10 years,1,5) soaring demand for lithium metal has meant that the global supply chain for lithium has had to grow nearly exponentially to keep up. The annual demand for lithium carbonate (Li2CO3) as a lithium metal has increased more than 3-fold compared with previous year in China.6) The present production of Li2CO3 is about half what would be needed to convert the 50 million cars produced every year into ‘plug-in hybrid electric vehicles’ (with an electric motor powered by a 7 kWh Li-ion battery and a combustion engine). However, the current LIBs rely on intercalation-based electrodes, that provide only limited practical energy density (~230 Wh·kg−1) in commercial electrical vehicles. Moreover, LIBs use non-aqueous, organic electrolytes that are prone to safety issues.1,3)
Sodium is one of the most naturally abundant elements and has low cost, low toxicity, and very suitable redox potential (Eo(Na+/Na) = −2.71 V versus standard hydrogen electrode; only 0.33 V positive of lithium). Sodium has physiochemical characteristics (iconicity, electronegativity, and electrochemical reactivity) similar to those of lithium. The ionic size of sodium ions is larger than that of lithium ions and Na has different bonding characteristics that have effects different from those of lithium systems. Most of the academic interest in Na-ion batteries has originated from the fact that similar materials (various crystal structures used in conventional Li-ion batteries) can be applied to Naion batteries.7) Na-ion batteries have gathered considerable interest in the field of large-scale energy storage systems (ESSs) because of the cost effectiveness and environmental friendliness.8-13) The early interest in sodium-based materials has been revisited on account of the natural abundance of sodium and relatively lower cost than of lithium resources. Therefore, rechargeable sodium-ion batteries (NIBs) can be considered for utilization in applications requiring lower cost and less weight/volume limitation in terms of the need to go beyond the limits of Li-ion batteries. Furthermore, from the perspective of articulation of Li and Na systems, there are no obstacles to research on Na-ion batteries because their binders, electrolytes, additives, etc., are similar. Inspired by these points, creation of an intercalative material applicable for Na-ion crystal structures has become attractive due to their potential for working well in sodium ion batteries. Recent attention to NIBs has led to many new designs for electrode materials being reported and proposed.14,15)
In order to construct a high performance Na-ion full cell, extensive investigation of electrode materials for sodium ion batteries has to be carried out not only various transition metal oxides and phosphates as cathode materials, but also candidate anode materials.16) Graphite has remained the most commonly used anode for Li-ion batteries since Sony and Asahi Kasei released the first commercial LIBs. However, the use of graphite results in poor electrochemical properties in sodium systems because sodium ions are nearly 25% larger than lithium ions.17) The larger size makes it more difficult for sodium ions to be inserted into the graphene sheets. Hard carbon showed better electrochemical performance than conventional graphite anode in sodium systems with 0.3 V of average operating voltage and 250 mAh·g−1 of reversible discharge capacity.18) However, its energy density is too low for it to be used as anodes for large-scale energy systems. Transition metals (e.g., Sn, Sb, Ge, Si, Pb) based on alloy reactions ensure considerable capacity as well as a relatively low redox potential because of their various oxidation states. Unfortunately, these types of materials undergo severe capacity fading due to the pulverization caused by large volume expansion during cycling.16,19)
Several types of transition metal oxides (TMOs), transition metal chalcogenides (TM-Chs; TM-S, Se, Te), and transition metal phosphides (P) based on conversion reactions have been reported for use as anode materials for NIBs.20) TMOs and TM-Chs, in particular, have advantages in terms of cost effectiveness, because they can be prepared easily via various synthesis processes (e.g., solid state reaction, high energy ball milling, spray pyrolysis, hydro/solvo-thermal synthesis, chemical vapor deposition, gas phase reaction, coprecipitation, electro-plating, electro-spinning, sulfurization, selenization). They also deliver high specific capacity comparable to transition metal-based electrodes. However, only a few TMOs in sodium-based systems have been able to utilize their high theoretical specific capacity because of their irreversibility for charge/discharge processes. The reaction mechanisms of both TMOs and transition metal chalcogenides (TM-Chs) are known to involve Na+ insertion into host materials with subsequent formation of sodium chalcogenide (NaxCh, x ≤ 2) and sodium-transition metal alloys. At the beginning of the discharge process, the conversion reaction between TM-Chs and Na+ is assumed to generate the transition metal and sodium chalcogenide (NaxCh, x ≤ 2). Successive reactions make up the alloying process, which transforms transition metals to sodium-transition metal alloys. These correspond to a conversion reaction and subsequent alloying reaction, respectively.
Moreover, the research on the anode materials for Na-ion batteries is still challenging. There remain problems that must be solved to improve the reversibility of sodium ion flow during electrochemical reactions. Selection of anode materials adequate for use in NIBs should provide low redox potential, high specific energy, good Na+ kinetics, and cycling stability. The most common strategies used to meet these requirements are morphology design, the introduction (or modification) of carbonaceous material, and the optimization of cell components (e.g., electrolyte, binder, additives). Remarkable research in the field of TM-Ch has led to advances in terms of productivity, and to excellent electrochemical performance.
2. Alloy-type Anode Materials
Alloy-type anodes for NIBs have been extensively reported by a number of authors over the last few years.19,21) Alloy anodes (Si, Ge, Sn, Pb, Sb, and P) have been attractive candidate materials for use in NIBs due to their high theoretical capacities as shown in Fig. 1(a). The general alloying reaction for electrochemical sodiation is shown in reaction (1).
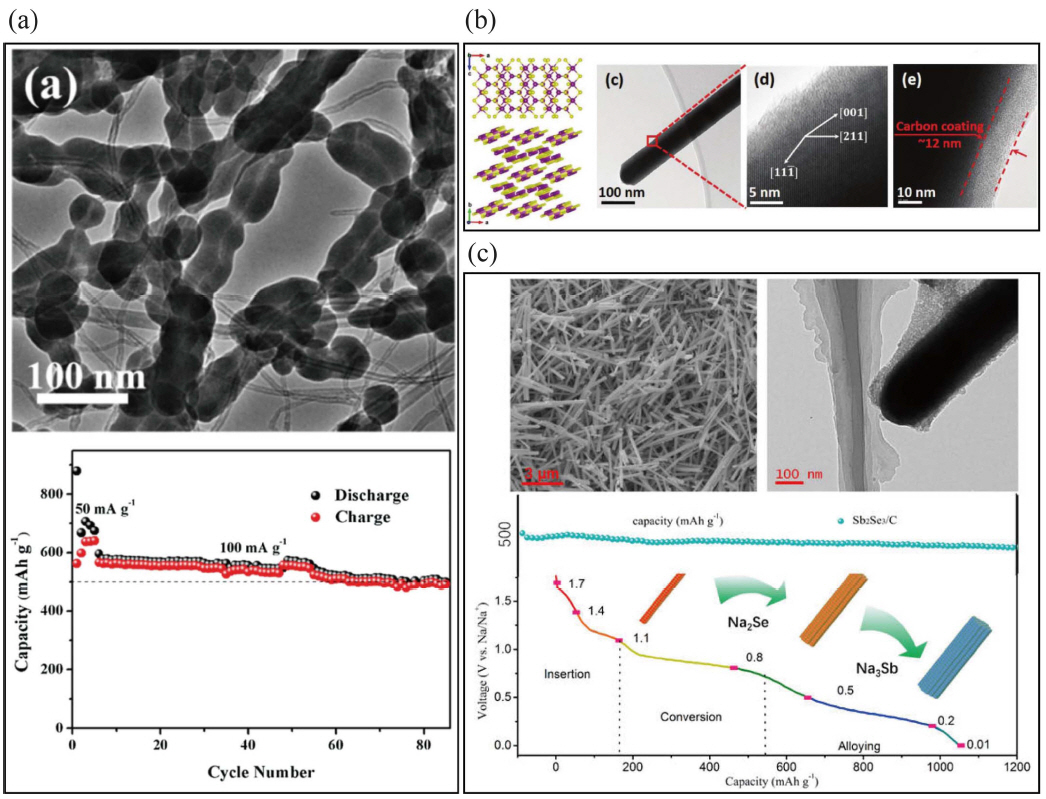
The alloying mechanism of each transition metal has been considerably studied using various methods by many groups. Komaba et al. examined the redox reaction of Sn-polyacrylate electrodes in aprotic Na cells with different types of binder, and then confirmed that Sn was transformed into a crystalline Na15Sn4 phase after full discharge using ex situ X-ray diffraction (XRD).22) Huang et al. studied phase transformation, which is associated with volume expansion, during electrochemical sodiation using in situ transmission electron microscopy (TEM) as shown in Fig. 1(b). The microstructural change from NaxSn (x ~ 0.5) to the final Na15Sn4 corresponded to (60 and 420) % volumetric expansion in accordance with the sodiation process. The final equilibrium phase for a Na-Sn alloy is Na15Sn4, which exhibits a high theoretical capacity (847 mAh·g−1) because of its high number of valence electrons.23) Ceder et al. showed the sodiation voltage curves obtained from the density functional theory (DFT) total energies for the operating voltage of the group IVa elements Si (954 mAh·g−1), Ge (369 mAh·g−1), Sn (847 mAh·g−1), and Pb (485 mAh·g−1), and reported voltage profiles below 0.5 V.19) Germanium has also been extensively studied as a negative electrode material for LIBs because of its high lithium storage capacity until the electrode crystallizes into Li15Ge4 (1384 mAh·g−1) and its high lithium diffusivity.24,25) The dissimilarity between Li and Na ions results in several orders of magnitude slower sodium diffusion than that of lithium in germanium. When sodium ions diffuse to interstitial sites in a germanium lattice, the larger radius of sodium (relative to lithium) requires much higher activation energy for hopping (0.51 eV for lithium, 1.5 eV for sodium). Loic et al. first reported the electrochemical properties of germanium thin-film electrodes during Na-ion electrochemical reaction. To investigate sluggish Na diffusivity into the interstitial sites in the lattice, the reaction potential data obtained with constant current and quasi-equilibrium measurements using the galvanostatic intermittent titration technique (GITT) are discussed and compared to X-ray absorption spectra (XAS) results.26,27)
In spite of their high theoretical capacity, more study of transition metals is required to reduce large volume expansion (which results in pulverization) by transformation to Na-TM (Si, Ge, Sn, Pb, Sb, and P) alloys. Especially, the volume expansion of Na-Sn alloys is about five times with respect to initial volume of tin as shown in Fig. 1(c). It has been reported that the deterioration of material properties and shortened cycle life due to pulverization are serious problem in long-time cycling tests.24,28,29) It causes a condition in which the electrode material and current collector separate from each other, which is associated with fatal fading of capacity. To solve this problem, a variety of studies are being conducted. These include such as physically preventing volume expansion using a carbon matrix,30,31) or minimizing the strain-stress that accompanies volume expansion using nano-sizing.32,33) Recently, there has been much research involving such as Na-TM alloys and matrix composites to reduce pulverization of transition metals. However, so far, no one has demonstrated perfect suppression of the volume expansion of transition metal during sodiation and de-sodiation, with reversible theoretical capacity.
3. Conversion-alloying Anode Materials
3.1. Transition metal oxides
Transition metal oxides can react with Na+ through a sequential conversion-alloying reaction. The conversion reaction between metal oxide and sodium takes place during the first discharge, generating sodium oxide and transition metal as shown in reaction (2). If the alloy anodes (Si, Ge, Sn, Pb, Sb, and P) remaining after the conversion reaction can be alloyed with sodium ions, they form a transition metal-sodium alloy as in reaction (3).
The remaining sodium oxide does not participate in successive cycles if the sodium oxide cannot secure the reversibility of Na diffusion. Problems with metal oxides have been pointed out, and these include such as the intrinsic irreversibility of the material and volume expansion (similar to alloy-based materials), which accompanies severe capacity fading during sodiation.
3.1.1. Tin oxide
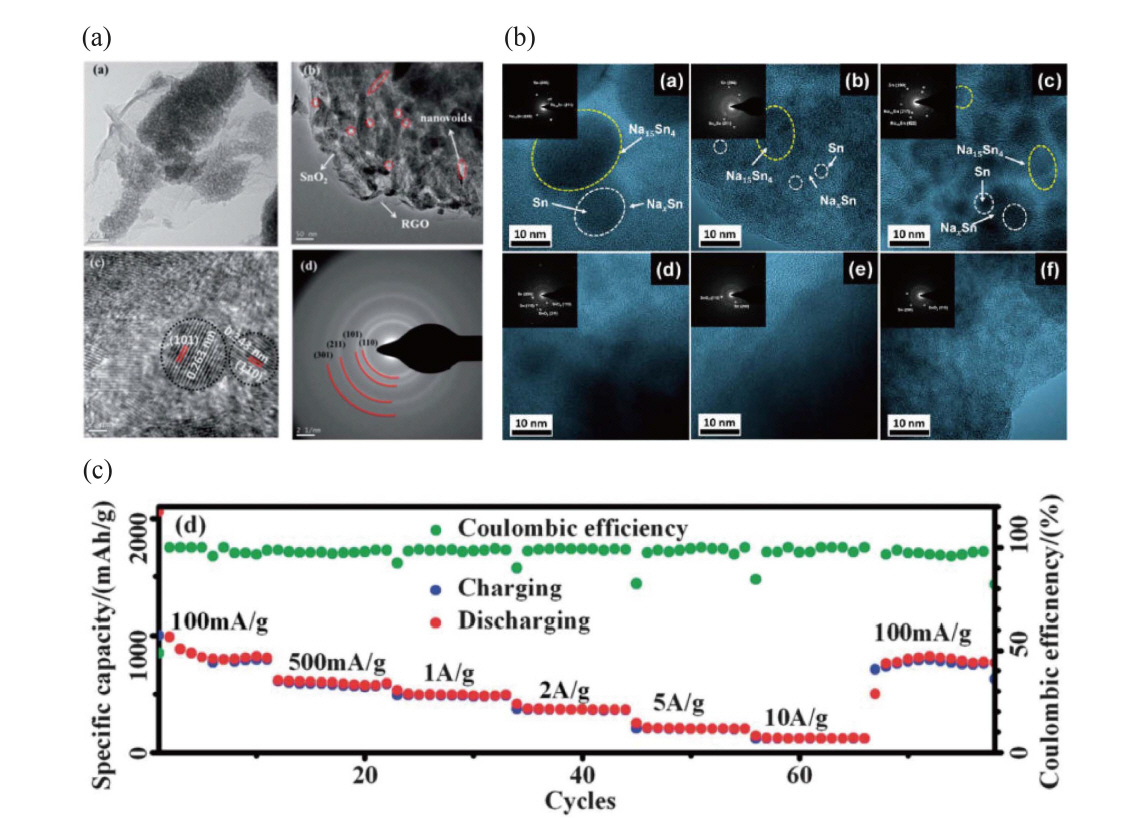
Various transition metal materials can achieve high capacity by an alloying reaction with sodium ions. For example, Sn reacts with 3.75 sodium atoms to provide a capacity of 1047 mAh·g−1, which is the highest among all the alloy anodes (except phosphorus). However, these transition metal materials exhibit a high volume expansion (4.2 times or more) of conventional Sn during the alloying reaction with sodium. This causes not only physical binding between the electrode material and the current collector, but also causes high strain-stress inside the material during charge/discharge reactions. As a result, it has been reported that the characteristic deterioration of life time due to pulverization remains a serious problem in long-term cycling tests. To solve this problem, a variety of studies are being conducted that include such as physically preventing volume expansion using a carbon matrix or minimizing strain-stress due to volume expansion using nano-sizing. Transition metal oxide-carbon composites have been reported as the common method to relieve volume expansion during the cycling, but also reduce the specific capacity because of the unnecessary carbon content. To try to overcome this shortcoming, Wang et al. reported the size of SnO2 nanoparticles plays a critical role in electrode performance as shown in Fig. 2(a).34) SnO2 nanoparticles (~ 5 nm) anchored on an rGO framework offers a buffering space and delivers high specific capacity (480 mAh·g−1 at 50 mA·g−1; 0.1 C-rate) and good rate capability (125 mAh·g−1 at 1000 mA·g−1; 2 C-rate). Fig. 2(c) shows that Song et al. aimed to form a complex between SnO2 and graphene prepared using facile hydrothermal synthesis.35) Tin dioxide can be well distributed on graphene oxide sheets when aided by dispersive hydrophilic radicals. This nanostructured composite exhibits not only better cycle performance but also better rate performance, with a long lifespan (> 1000 cycles). Patra et al. synthesized 1 nm SnO2 particles uniformly dispersed and tightly connected to carbon supports.36) This arrangement provided an initial charge-capacity of 400 mAh·g−1 at 100 mA·g−1 and capacity-retention of 82% after 100 cycles. The lower than theoretical capacity of the 1 nm SnO2 nanoparticle electrode can be explained by an incomplete alloying reaction because of the lower diffusivity of Na in Sn. In addition, in a number of studies there are reports of introducing carbon supports such as carbon additives, carbon nanotubes, and graphene nanosheets to solve the low intrinsic electron conductivity problem of SnO2; however, none could approach the theoretical capacity in Fig. 2(b).37-40) Xie et al. reported the fabrication of cross-linked 3-D conductive graphene networks with hierarchical pores and SnO2 nanoparticles. This functional structure provides an efficient electron pathway within the graphene networks, but shows lower reversible capacity (< 400 mAh·g−1) because of the high carbon content in the electrode material.38) Huang et al. also reported a 3-D porous carbon encapsulated SnO2 nanocomposite synthesized using an in situ methodology that delivered a reversible specific capacity of 280.1 mAh·g−1 after 250 cycles at a current density of 100 mA·g−1.39) Although it exhibited 100 mAh·g−1 after 100 cycles at a high current density of 1600 mA·g−1, the reversible capacity did not reach the theoretical capacity, as in other publications about the use of SnO2 nanocomposite electrodes for NIBs.
3.1.2. Antimony oxide
Antimony (Sb) reacts with three sodium ions and has a theoretical capacity of 660 mAh·g−1, which is relatively smaller than that of tin (847 mAh·g−1). However, the process of sodiation and de-sodiation of antimony (Sb) can lead to longer lifetimes during long-term cycling tests than that of tin (Sn) because of the lower volume expansion (~ 3 times) during the alloying reaction than that of tin (4.2 times).41) To date, tin oxides (SnO and SnO2) has been extensively studied for the purpose of improving lifetimes by suppressing the volume expansion accompanying the charge/discharge reactions of Sn with Na. As the same strategy, a variety of research has been conducted to increase the reversible specific capacity of antimony oxides (Sb2O3 and Sb2O5). Anti-mony oxides (Sb2O3 and Sb2O5) react with 10 and 14 sodium ions per mole, respectively, thus exhibiting a theoretical specific capacity of about (919 and 1236) mAh·g−1.
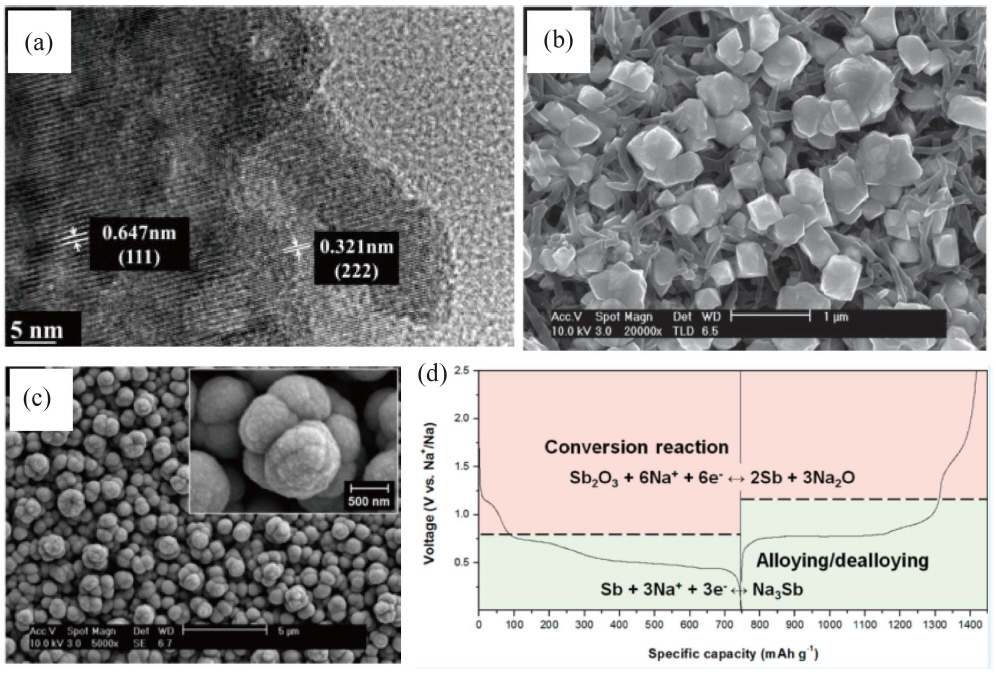
Hu et al. reported combined conversion-alloying electrochemical reactions of Sb2O3, delivering a reversible charge capacity of 509 mAh·g−1 at a current density of 0.05 A·g−1, and retains as high as 265 mAh·g−1 at an ultrahigh current density of 5 A·g−1 as shown in Fig. 3(a). Porous 3-D Sb-Sb2O3-Polypyrrole (PPy) nanocomposites were fabricated using a facile electrodeposition method with a PPy nanowire network as shown in Fig. 3(b).42) They exhibited a reversible charge capacity of 512.01 mAh·g−1 over 100 cycles, and outstanding rate capability (299 mAh·g−1 at 3300 mA·g−1, 56.7% of the charge capacity). Although the Sb-Sb2O3-PPy electrode exhibited excellent electrochemical properties, the reversible capacity was lower than the theoretical value of a metallic Sb electrode due to its carbon content (~ 15%). Fig. 3(c) shows that Sb-Sb2O3 composites using a one-step electrodeposition process in an aqueous electrolytic bath.43) The morula-like Sb-Sb2O3 composites delivered a reversible capacity of 670 mAh·g−1, which is beyond the theoretical capacity of Sb (660 mAh·g−1). These composites were of metallic Sb and crystalline Sb2O3 in a molar ratio of 9 : 1. The high specific capacity resulted from the reversible formation of Na2O and Sb2O3 that occurs in addition to the alloying reaction of Sb with Na as shown in Fig. 3(d). In many of the studies in the literature graphene oxide was introduced to find appropriate agreement between the capacity increase of antimony oxide and the improvement of the rate characteristics. Wang et al. synthesized a Sb2O4-reduced graphene oxide (rGO) via solvothermal synthesis with different graphene oxide content in the nanocomposite.44) The Sb2O4-rGO nanocomposite containing 18 wt% of graphene oxide showed a rechargeable capacity of 551 mAh·g−1 after 100 cycles and a rate performance of 401 mAh·g−1 at a current density of 1 A·g−1. However, there was still an obstacle to its use in development of high-performance NIB anodes because of the intrinsic properties of the antimony oxide-based materials. Fei et al. reported a flexible Sb2O3-carbon cloth (CC) composite used as a freestanding anode, which was fabricated using a simple solvothermal method.45) This electrode delivered 900 mAh·g−1, which is very close to the theoretical capacity of antimony oxide (Sb2O3), after 100 cycles at a low current density of 50 mA·g−1. The outstanding electrochemical performance was attributed to the homogeneous growth of Sb2O3 nanoparticles on the CC, which improved electronic migration, sodium ion diffusion, and structural stability during the conversion and alloying reactions.
3.1.3. The other transition metal oxides
A number of candidate anode materials based on a sodium insertion host for SIBs have been intensively studied due to their low cost, nontoxicity, low operation voltage, low strain, and excellent cyclability.19,21) Generally, titanium-based materials deliver low specific capacity due to the limitation of alkali-ion storage sites. Spinel Li4Ti5O12 is one of the most attractive materials for a so called “zero-strain” anode for LIBs. It was first reported and further studied as an anode for NIBs by Zhao and Sun.47,48) The Li4Ti5O12 electrode can also store sodium with an average voltage of 0.91 V, delivering a reversible capacity of 155 mAh·g−1, and offers the best stable cyclability. A three-phase separation mechanism (2Li4Ti5O12 + 6Na+ + 6e− ↔ 2Li7Ti5O12 + Na6LiTi5O12) of the electrode, with respect to sodium diffusion, can be predicted by density functional theory (DFT) calculations, which results have been confirmed through in situ synchrotron XRD and advanced TEM techniques. Tarascon and Palacin’s group first ever reported that Na2Ti3O7 reversibly reacted with sodium as a low average potential of 0.3 V (demonstrating 200 mAh·g−1) to reversibly uptake two Na ions per formula unit.49) P2-type layered Na0.68[Li0.22Ti0.78]O2 delivered a reversible specific capacity of 116 mAh·g−1 (corresponding to 0.38 Na+ ion insertion per formula unit) at an average storage potential of 0.75 V. It showed an ultra-long cycle life with 75% capacity retention, and confirmed that the volume change during Na+ insertion/extraction was only 0.77% (zero strain) after 1200 cycles.50) Recent progress on Ti-based sodium insertion electrodes has provided opportunities for the development of large-scale stationary energy storage systems.51) Most of the intercalative materials based on sodium titanates such as Na2Ti3O7, Na2Ti6O13,52) Na0.68[Li0.22Ti0.78]O2, and sodium nonatitanate53) have been investigated for creation of stable anode materials with low operating voltages, but their practical capacity did not meet expectations for the energy storage applications of the future.
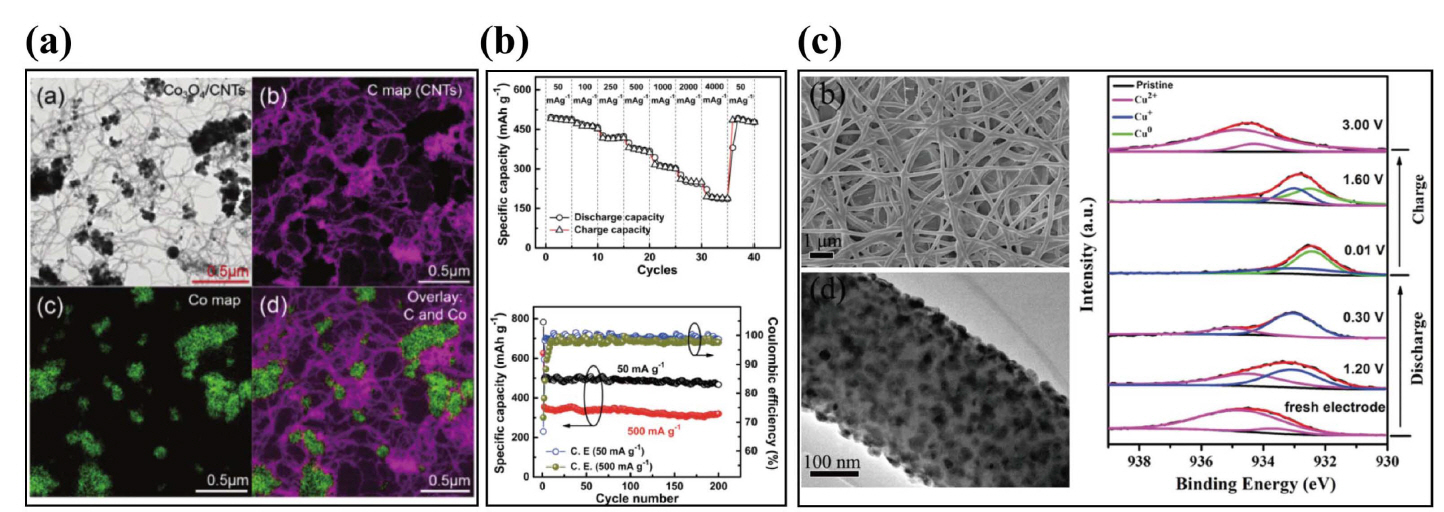
Recently, many metal oxides and sulfides have been studied as materials for negative electrodes for NIBs since the transition metal oxide spinel NiCo2O4 was first reported as an anode for NIBs in 2002.54) NiCo2O4 undergoes a phase transformation to metallic Co and Ni after full sodiation, which was demonstrated by ex situ XRD. NiCo2O4 delivered a reversible capacity of 200 mAh·g−1, which was much lower than its theoretical capacity (890 mAh·g−1). This is the first time that a reversible conversion reaction was found for the mixed spinel composite, thereby providing an alternative anode material and a new approach for Na-ion batteries. Inspired by this electrode material, there have been studies reporting various combinations of metal oxides and sulfide as anodes for NIBs.55-62) Iron oxides (Fe2O3 and Fe3O4) are attractive anodes due to their high theoretical capacity, easy synthesis, low toxicity, and low cost.56,57,60) Nano-crystallized Fe3O4 (10 nm) prepared by the precipitation method, had a demonstrated reversible capacity of 160 - 170 mAh·g−1 with excellent capacity retention. The particle size of Fe3O4 has an effect on its structural change and electrochemical activity, which was confirmed by ex situ XRD for the cycled electrode samples.56) In terms of cost effectiveness, Fe3O4 is one of the most promising potential anode materials for sodium-ion battery systems. A full NIB cell based on Na[Ni0.25Fe0.50Mn0.25]O2-Fe3O4/C with an electrolyte (NaClO4 in EMS/FEC) was investigated, and delivered a reversible capacity of 130 mAh·g−1 and 76.1% capacity retention after 150 cycles.63) Similarly, Kuma et al. reported the synthesis of pure Fe3O4 nanoparticles via the hydrothermal method, and investigated their electrochemical performance with different binder systems (PVDF and Na-alginate).57) Fe3O4 nanoparticles with the alginate binder delivered stable capacity of 248 mAh·g−1 at 0.1 C-rate after 50 cycles. The Na-ion full cells in which a Fe3O4-alginate anode and Na3V2(PO4)3-graphene cathode were combined, confirmed the potential to apply these directly to realize low-cost NIBs in the future. Many anodes of these types have been studied extensively, including CoO, CuO, NiO, MnO2, and MoO3 for Na-ion battery systems as shown in Fig. 4.55,58,59,61,62) The electrochemical performances of transition metal oxide as an anode for sodium-ion batteries are summarized in Table 1.
3.1.4. Limitation of transition metal oxide anodes
Transition metal oxide can be expected to have high specific capacity as anode materials for sodium ion batteries due to their variety of oxidation states, but they have poor rate performance owing to the extremely low intrinsic electrical conductivity of the electrode. Moreover, the sluggish diffusivity of sodium ions in the transition metal oxides limits the rate of sodium insertion/extraction (low energy density), and the low surface area of the bulk electrodes restrains their capacitive contribution (low power density). To solve these problems, many studies have been carried out to improve the electrical conductivity by introducing a carbon matrix and composite, and making short diffusion paths by limiting the particle size to nanometer-scale to enhance the ionic conductivity. However, these noteworthy attempts were insufficient to realize the necessary performance of sodium for high-efficiency energy storage. If a carbon material is applied to overcome the intrinsic properties of the transition metal oxides, the weight ratio of the oxides is remarkably decreased as the fraction of carbon matrix in the electrode material is increased. Furthermore, various problems are encountered in the packaging process as well as the practical capacity in actual battery applications. The rational design of controllable microstructure and electron conductive components of transition metal oxides for high-performance NIB anode materials is both necessary and urgent.
3.2. Transition metal chalcogenides
Transition metal chalcogenides are chemical compounds consisting of at least one chalcogen anion and at least one more transition metal cation. Transition metal chalcogenides have attracted a great deal of interest as potential anode materials for NIBs because of their unique crystal structure, which consists of 2-D or quasi 2-D stacked layers like graphene nanosheets.66,67) They guarantee a certain reversibility and conductivity because of their lower bonding energy. This difference in their discharge/charge characteristics can be attributed to the relative reversibility of the formation of sodium chalcogenides (NaxCh; Ch = S, Se, and Te) compared to the formation of sodium oxides (NaxO). The chalcogenide anion of the electrode material is converted to sodium polysulfide (Na2Chx; 2 ≤ x ≤ 8) during the conversion reaction, ultimately becoming sodium chalcogenide (Na2Ch). This conversion reaction applies equally to other chalcogen ions. After the conversion reaction, the remaining the transition metal can be alloyed with sodium ions like the reaction (3) if the transition metal is the alloy anodes (Si, Ge, Sn, Pb, Sb, and P). They form a transition metal-sodium alloy as in reaction (5). The platform reaction can be expressed as indicated in reaction (4) and (5) below.
Transition metal chalcogenides are considered strong candidates due to their unique physical properties. These include such as a tunable stoichiometric compositions, unique crystal structures, chemical properties such as rich redox sites, and relatively higher electrical conductivity, relative to their transition metal-oxide counterparts.97-99) The electrochemical performances of transition metal chalcogenide as an anode for sodium-ion batteries are summarized in Table 2.
3.2.1. Tin chalcogenides
Tin chalcogenides (SnChs) have been used in lithium ion batteries (LIBs) as promising anode materials. Because 1 mole of SnCh or SnCh2 could accommodate (5.75 or 7.75) moles of alkali ion+, they were able to deliver high theoretical capacity (1022 or 1136) mAh·g−1. The previous reports of tin sulfides as anode material for NIBs were only focused on improvement of battery performance by optimizing the electrode additives, using suitable electrolytes, and adjusting the content of graphene oxide.70,71) Understanding of the reaction mechanism during sodiation/de-sodiation is still lacking, which leads to incomplete electrochemical utilization for Na-storage systems due to irreversibility of unfavorable conversion-alloying reactions.
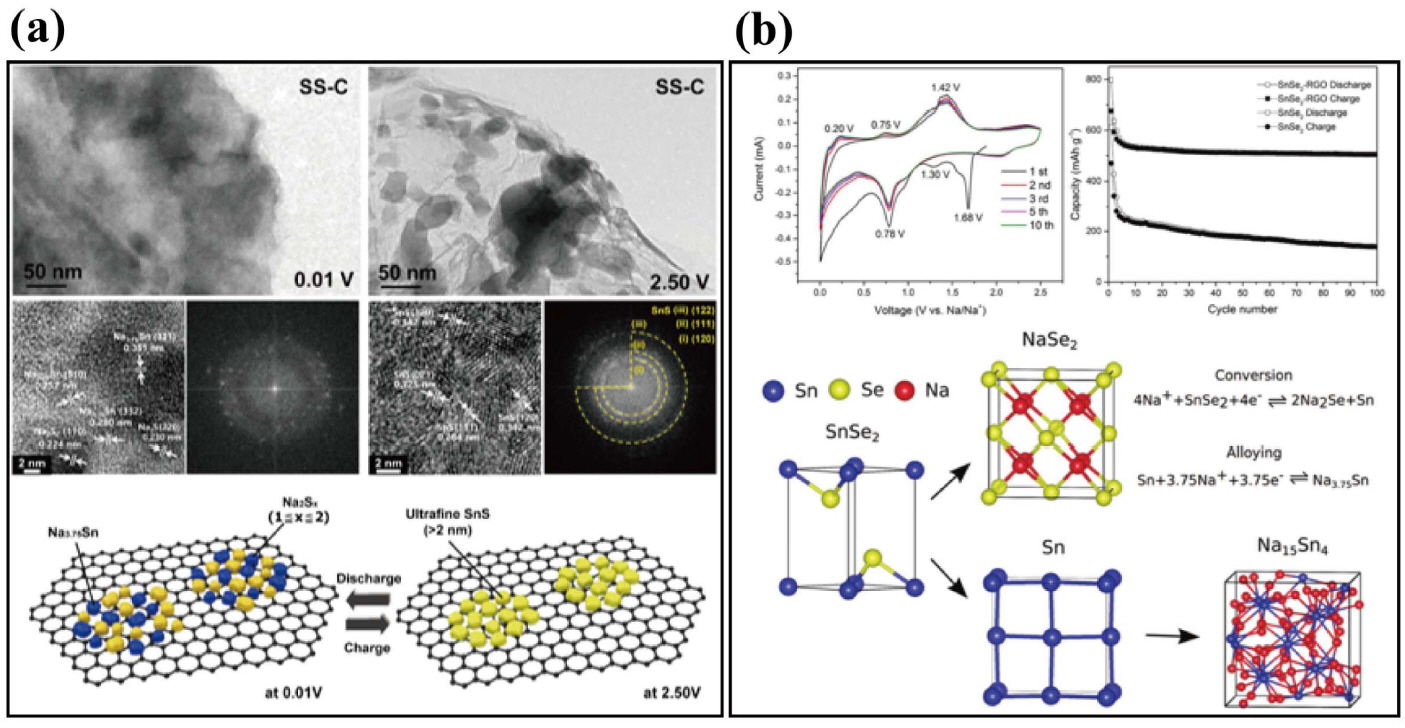
Zhou et al. studied the structural changes of a hybrid SnS@graphene architecture via hydrothermal synthesis during cycling.70) The electrode composite underwent fewer phase transitions after the conversion reaction than did SnS2@graphene hybrid material, as revealed by ex situ XRD. This material can provide high reversible capacity and superior high-rate capability allowing the introduction of inexpensive and versatile synthesis techniques. Kim et al. prepared mixed Sn-S nanocomposites uniformly distributed on reduced graphene oxide via facile hydrothermal synthesis and a unique carbo-thermal reduction process as shown in Fig. 5(a). The product was ultrafine nanoparticles with crystalline size of 2 nm.74) These nanocomposites have been experimentally confirmed to overcome the intrinsic drawbacks of tin sulfides such as large volume change (4.2 times), with accompanying cycle instability due to the pulverization problem, and sluggish diffusion kinetics. The mixed Sn-S ultrafine nanoparticles exhibit outstanding electrochemical performance: an excellent specific capacity of 1230 mAh·g−1 and an impressive rate capability (445 mAh·g−1 at 5000 mA·g−1). The electrochemical behavior of a sequential conversion-alloying reaction for the anode materials was investigated, revealing both the structural transition and the chemical state during the discharge/charge process. Kim et al. first examined SnSe-C composite, prepared via facile ball milling in Ar gas.75) The reversible capacity of the composite was 707 mAh·g−1, delivering 91% of the theoretical capacity of SnSe (780 mAh·g−1). This calculation was based on the weight of SnSe alloy in the composite. The reversible sodiation/de-sodiation mechanism of SnSe was determined through ex situ XRD and TEM, which contributed to the development of transition metal selenide materials as anode materials for NIBs. Other tin selenide materials such as 2-D SnSe2 alloy with rGO, were synthesized by a hydrothermal method in Fig. 5(b).77) The SnSe2-rGO nanocomposites delivered a reversible specific capacity of 515 mAh·g−1 at 0.1 A·g−1 after 100 cycles. The rGO is believed to stabilize the electrode, improve electron conductivity, and better tolerate volumetric changes during the sodiation/de-sodiation processes. Park et al. reported a difference in the electrochemical properties of cubic crystal-structured SnTe for Li and Na-ion battery anodes.78) The electrochemically induced phase transition mechanism of SnTe electrodes during Li and Na insertion/extraction was investigated using various ex situ analysis techniques. SnTe was converted to Li4.25Sn and Li2Te during lithiation; however, during Na insertion, SnTe experienced a quasi-intercalation reaction to NaxSnTe (x ≤ 1.5) and conversion to Na3.75Sn and Na2Te.
3.2.2. Antimony chalcogenides
The electrochemical reaction mechanism in the sodiation/de-sodiation process of antimony chalcogenides has a conversion and alloying reaction principle similar to that of the tin chalcogenides. Zhu et al. prepared a flower-like Sb2S3 composite via a polyol reflux process that delivered reversible capacity of 835.3 mAh·g−1 (50 mA·g−1 after 50 cycles), near the theoretical capacity (946 mAh·g−1) of Sb2S3.79) They insisted that such an improvement of the electrochemical performance was attributable to the unique flower-like structure of Sb2S3. This composite not only exhibited mitigated volume expansion, but also provided effective diffusion pathways for electrons and Na ions. Kim et al. also synthesized cystalline Sb2S3 (c-Sb2S3) and amorphous Sb2S3 (a-Sb2S3) nanoparticles via a similar polyol synthetic protocol.80) The reversible charge capacity of a-Sb2S3 was 512 mAh·g−1 after 100 cycles with a capacity retention of about 79% (vs the first charge capacity). The c-Sb2S3 nanoparticle electrode after 100 cycles exhibited poor electrochemical performance in the cycling and rate tests, although the initial coulombic efficiency of c-Sb2S3 was similar to that of a-Sb2S3. These were beneficial for reducing the large volume of expansion during sodiation and for enhancing electrical contact. Antimony sulfide (Sb2S3) anode materials contained added carbon introduced to provide improved electron paths and stable reaction sites have been reported. These exhibit high reversible specific capacity and stable lifetime characteristics as shown in Fig. 6(a), (b).82-85)
Since Jiang et al. found that Sb2Se3@C prepared via high-energy mechanical ball milling of Sb, Se, and carbon black showed a reversible capacity of 650 mAh·g−1 at 0.1 A·g−1, antimony selenides and tellurides have become attractive as promising anode materials for NIBs as shown in Fig. 6(c).86-88,100) One-dimensional Sb2Se3/C nanorods grown toward the [001] direction show reversible capacity of 485.2 mAh·g−1 after 100 cycles and 311.5 mAh·g−1 at a current density of 2.0 A·g−1.87) Nam et al. synthesized the nanostructured Sb2Te3-C composites via solid-state reaction and a high-energy ball milling method to investigate the electrochemical properties of the nanocomposites. The Sb2Te3-C nanocomposite showed better electrochemical performance with long cycle life, relative to those of the Sb2Te3 electrode. The sodiation/de-sodiation mechanisms of the Sb2Te3-C nanocomposites, demonstrated using ex situ XRD and TEM, confirmed the occurrence of conversion/recombination reactions, whereas Sb2Te3 only displayed conversion/non-recombination reactions (no recovery of the original Sb2Te3 phase).
3.2.3. The other transition metal chalcogenides
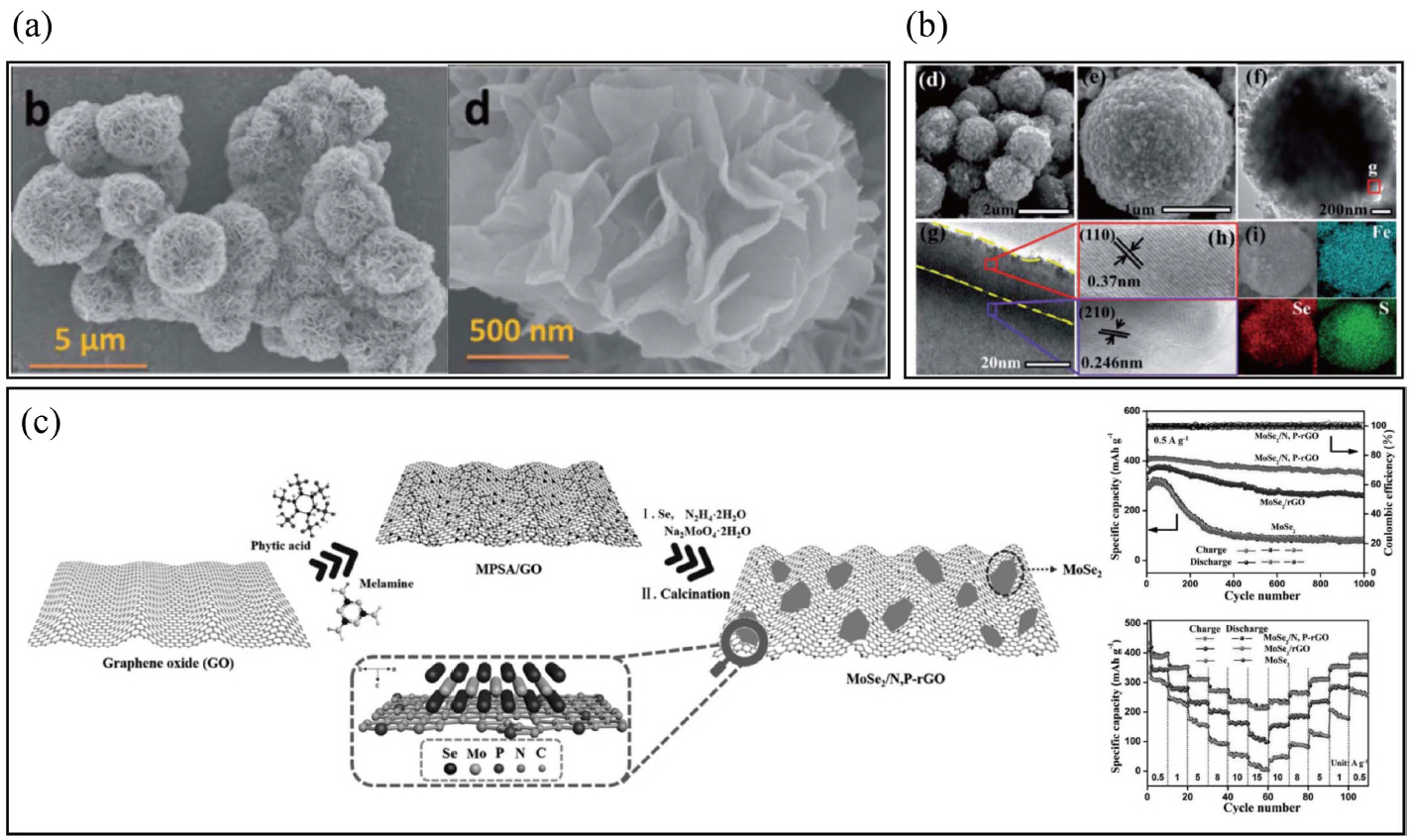
Many other layered transition metal chalcogenides also have been reported for use as anodes for NIBs as shown in Fig. 7.89-91,94-96) Molybdenum disulfide (MoS2), a typical layered material, has been intensively investigated as an anode for NIBs. Park et al. first studied layered MoS2 as an intercalation host for room temperature NIBs, although the Na/MoS2 cell had a discharge capacity of 85 mAh·g−1 after 100 cycles, with poor cycle retention. The discharge reaction was interpreted as occurring in two steps: xNa + MoS2 → NaxMoS2. The first step is the intercalation step (x ≤ 0.5) and next is the conversion step (0.5 ≤ x ≤ 1.1).101) Layered-structure MoSe2 has a large interlayer distance (~ 0.65 nm), small band gap (~ 1.1 eV), and a theoretical capacity (~ 422 mAh·g−1) that makes MoSe2 promising for use in NIBs. However, there are serious challenges from the low electronic conductivity, large volume change, and high mechanical stress/strain during sodiation/de-sodiation. To solve these problems, MoSe2 homogenously distributed on N,P-doped carbon nanosheets was synthesized using a solvothermal method, followed by high-temperature calcination.91) This combination presented excellent cycle stability of 378 mAh·g−1 after 1000 cycles at 0.5 A·g−1 with 87% cycle retention (vs. the second cycle), and rate capability (the specific capacity at 15 A·g−1 equals ~ 55% of that of 0.5 A·g−1). Several non-layered iron sulfides and nickel sulfides for negative electrode materials have been reported, such as FeS2 and Ni3S2.92,93,102,103) Zhao et al. synthesized lychee-like FeS2@FeSe2 core-shell microspheres, which delivered a reversible discharge capacity of 350 mAh·g−1 at 1 A·g−1 after 2700 cycles, and 301.5 mAh·g−1 at 5 A·g−1 with a coulombic efficiency of 97%.93) Such a transition metal chalcogenide electrode had excellent cycle stability and superior rate capability; however, these types of transition metals cannot alloy with sodium, so their reversible capacity is too low to apply for use in high energy-density storage systems.
Recently, germanium (Ge) has become attractive as an anode material for NIBs because of previous reports of its high lithium diffusivity and high lithium storage capacity (until the electrode crystallizes into Li15Ge4) of 1384 mAh·g−1.24,104) Chevrier et al. calculated the sodiation voltage curves obtained from the DFT total energy for germanium, and got average sodiation voltages of < 0.4 V (vs Na/Na+). Abel et al. synthesized nano columnar germanium thin films by evaporative deposition that had high discharge capacity (403 mAh·g−1 ≈ that of amorphous Na1.17Ge).105) This exceeded the theoretical value (369 mAh·g−1) of the fully sodiated NaGe phase). Kim et al. first reported germanium disulfide (GeS2) nanocomposites uniformly distributed on reduced graphene oxide as a promising anode material for NIBs. These were prepared via facile hydrothermal synthesis and a unique carbothermal annealing technique.96) The GeS2 hybrid anode yielded a high reversible specific capacity of 805 mAh·g−1 (> theoretical capacity), an excellent rate capability of 616 mAh·g−1 at 5 A·g−1, and a cycle retention of 89.4% after 100 cycles. A combined ex situ characterization study revealed that electrochemically driven amorphization plays a key role in achieving efficient sodium storage by accommodating excess sodium ions in the electrode materials. Understanding the sequential conversion-alloying reaction mechanism for GeS2/rGO hybrid anodes should provide a new approach for developing high-performance energy storage applications.
3.3. Transition metal phosphides
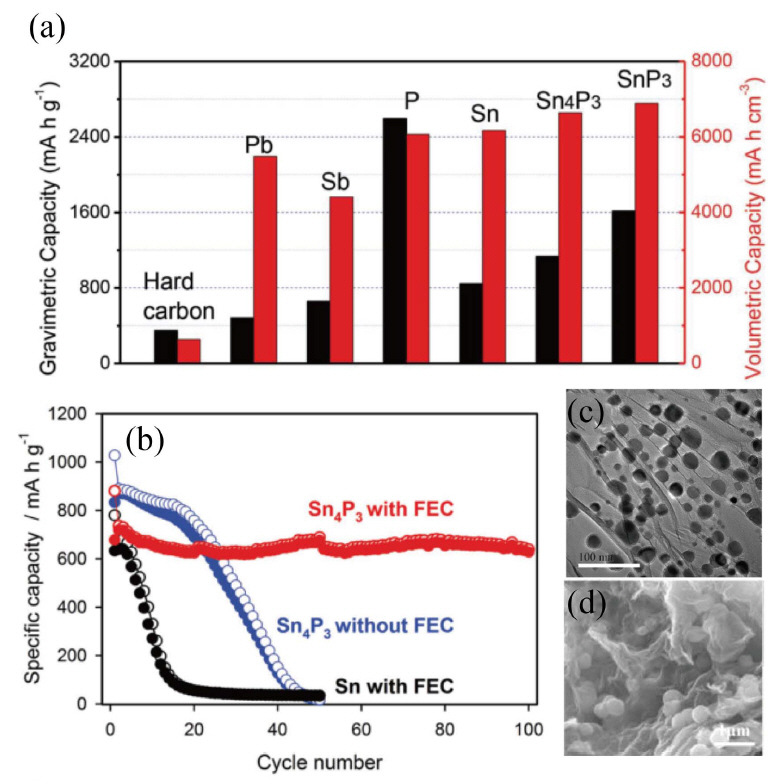
Phosphorus (P) is one of the most promising anode candidates, and studies have demonstrated high theoretical specific capacity of 2596 mA·h−1 corresponding to final phase Na3P.106,107) The operating voltage in NIBs appears to fall in the range 0.0 - 0.5 V, which guarantees high energy density as well as high reversible capacity as shown in Fig. 8a. The platform reaction of phosphorus can be expressed as in reaction (6) and (7) below.
Phosphorus is a Group V non-metallic element in the periodic table and has three allotropes known as white, red, and black phosphorus. White phosphorus begins to ignite in air at 30°C, so it is difficult to use as electrodes and not suitable in terms of safety. To date, red and black P and their composites have been extensively studied as anode materials because of their chemical stability at room temperature and atmospheric pressure. The characteristics of phosphorus anodes for NIBs include 1) high electrochemical performance, 2) cost effectiveness, 3) stability in air (but oxidation in air with a low ignition temperature with white phosphorus, 4) large volume expansion during sodiation, 5) acceptable diffusivity and conductivity of each step (intercalation and alloying), and 6) the need for an in situ method to measure the conductivity of intermediate states (Na3P→ Na2P→ NaP→P). To solve these problems and allow phosphorus to be utilized as anode material for NIBs, red phosphorus-CNT hybrid nanocomposites were synthesized by simple mixing.125) These materials provided a reversible capacity of 1675 mAh·g−1 with capacity retention of 76.7% over 10 cycles. This suggest that simple synthesis has the potential to allow utilization of red P-CNTs as a promising anode candidate for NIBs with high reversible capacity and low cost. Li et al. synthesized red P@CMK-3 hybrid material by melting-diffusion.126) To avoid the ignition of commercial phosphorus, all synthesis processes were performed in inert gas. Subsequent treatment was heating at 450°C in a sealed vessel for 3 h, after which it was cooled to 260°C for 24 h to convert white P to red P. For the phosphorus-carbon matrix composite, this accommodate large volume change (~ 3 times) of the phosphorus during sodiation/de-sodiation and offered high electron conductivity. However, the reversible capacity based on the mass of red P was only 1020 mAh·g−1 after 210 cycles at 5 C-rate (If calculated based on the mass of red P-carbon composite, the reversible capacity was only 370 mAh·g−1; P = 31.54 wt%).
Kim et al. first reported ball milling of tin phosphide (Sn4P3) for use as a NIB anode. Sn4P3 demonstrated excellent electrochemical performance including a reversible capacity of 718 mAh·g−1 and stable cycling with negligible capacity fading over 100 cycles as shown in Fig. 8(b).127) Inspired by this report, many tin-based phosphide nanocomposites have been extensively studied for use in NIBs.108,112-114,128,129) Huang et al. prepared molybdenum phosphide (MoP) nanorods wrapped with a thin carbon layer and disclosed an electrochemical reaction mechanism utilizing in situ X-ray diffraction. The nanocomposites exhibited a remarkable rate capability (104.5 mAh·g−1 at 1600 mA·g−1 even after 10,000 cycles), but the reversible discharge capacity was only 398.4 mAh·g−1 at 100 mA·g−1 after 800 cycles. Wu et al. have developed an assembly and self-tem-plate strategy for the fabrication of Ni2P@GN hybrids with 3D yolk-shell-like architecture as shown in Fig. 8(c), delivering a high charge capacity of 511 mA h·g−1 after 250 cycles with a capacity retention of 93%.110) CoP nanoparticles uniformly impregnated in N-doped carbon nanosheets were synthesized using a facile calcination method and delivered a reversible capacity of 598 mAh·g−1 at 100 mA·g−1.120) Amorphous Se4P4 is promising as anode material, and was synthesized by a mechanical milling technique. It exhibited a high reversible capacity of 1048 mAh·g−1, corresponding to 86% of the theoretical capacity (1217 mAh·g−1) at 50 mA·g−1.124) The full cell battery (Se4P4 anode and Na3(VO0.5)2(PO4)2F2/C cathode) delivered a reversible discharge capacity of 985 mAh·g−1 based on the mass of Se4P4 with an average output voltage of ~ 2.5 V for the first two cycles. Many other transition metal phosphides such as Cu3P, CuP2, CoP, FeP, and Ni2P have also been revisited because of their potential for utilization in anodes for NIBs.110,111,117-123) The transition metal phosphide as an promising anode for sodium-ion batteries are reported as shown in Table 3.
4. Summary and Outlook
The qualities that must be provided in battery materials for future energy markets is low-cost, environmental compatibility, safety, and high performance for energy storage. Due to the increase in the amounts of time that small electronic devices are used and to the development of electric vehicles and large-scale energy storage systems, sodium ion batteries with high energy density are essential. The development of anode materials for sodium ion batteries to overcome the limitations of conventional lithium ion batteries can be expected to provide significant applications for renewable energy devices. The creation of nano-structured electrodes and carbon composites via various synthesis methods has been reported as a strategy to overcome the obstacles affecting safety, high cost, and ease of manufacturing large batteries, which is a vulnerable point of NIB anode materials. Through in-depth understanding of the electrochemical properties of high-performance electrodes and the in situ and ex situ analysis after charge/discharge processes, it is expected that the sodium secondary battery will not only play a pivotal role in energy related fields, but also will have a large effect on the high value-added technology of the future.