Jung, Jeon, Park, Jung, Myoung, and Yang: Growth Behavior of Thermally Grown Oxide Layer with Bond Coat Species in Thermal Barrier Coatings
Abstract
The effects of bond coat species on the growth behavior of thermally grown oxide (TGO) layer in thermal barrier coatings (TBCs) was investigated through furnace cyclic test (FCT). Two types of feedstock powder with different particle sizes and distributions, AMDRY 962 and AMDRY 386-4, were used to prepare the bond coat, and were formed using air plasma spray (APS) process. The top coat was prepared by APS process using zirconia based powder containing 8 wt% yttria. The thicknesses of the top and bond coats were designed and controlled at 800 and 200 μm, respectively. Phase analysis was conducted for TBC specimens with and without heat treatment. FCTs were performed for TBC specimens at 1121°C with a dwell time of 25 h, followed by natural air cooling for 1 h at room temperature. TBC specimens with and without heat treatment showed sound conditions for the AMDRY 962 bond coat and AMDRY 386-4 bond coat in FCTs, respectively. The growth behavior of TGO layer followed a parabolic mode as the time increased in FCTs, independent of bond coat species. The influences of bond coat species and heat treatment on the microstructural evolution, interfacial stability, and TGO growth behavior in TBCs are discussed.
Key words: Thermal barrier coating, Bond coat species, Furnace cyclic test, Thermally grown oxide, Growth behavior
1. Introduction
Thermal Barrier Coatings (TBCs) are widely applied to high temperature components of gas turbines, such as blades, vanes, and combustors to prevent thermal degradation of their base materials from thermal and mechanical stresses. 1,2) The typical TBC system has a layered structure with a Ni-base superalloy as a substrate, an MCrAlY as a bond coat (M = Ni, Ni-Co, and Co-Ni) which can protect the substrate from oxidization and corrosion, and a zirconia-based ceramic material as a top coat with a relatively low thermal conductivity and high thermal expansion coefficient. The top coat serves to enhance the thermal barrier effect of the entire system in high-temperature environments. Its bond coat prevents delamination caused by thermal expansion mismatch between the substrate and the top coat, enhances adhesion with the top coat, and protects the substrate from oxidation. TBC system, connected to the internal cooling channel system, decreases the temperature range to which high-temperature gas turbine components are exposed by over 150°C, allowing them to operate under their respective melting point temperature ranges. 3-5)
The TBC system is fabricated via the following methods: air plasma spray (APS), vacuum plasma spray, high-velocity oxy-fuel spray (HVOF), and electron beam physical vapor deposition (EB-PVD). Here, the HVOF method uses high-velocity flame with a velocity of over 800 m/s to melt and deposit feedstock powder, but is only applied to form a bond coat due to its relatively low flame temperature. The EB-PVD method uses an electron beam to form columnar structure, and is widely applied to form a top coat in aviation TBC systems. The APS method is extensively used because it is capable of forming both the top and bond coats. 6-10) Currently, in an attempt to improve the efficiency of the gas turbine system, its inlet temperature is being adjusted upward. In response, extensive relevant research is being conducted to improve the performance of the TBC system, such as thermal shielding performance and heat resistance. To this end, researchers are exploring effective ways to control critical factors including coat thickness, chemical composition, and microstructure, and address the problem of early delamination caused by internal defects and cracks forming in coating layers. 10-15)
The TBC system, generally applied to high-temperature components, is repetitively exposed to thermal and mechanical stresses during gas turbine operation, leading to failure when its top coat is delaminated. 3,16,17) Such failures are mainly attributed to the formation of thermally grown oxide (TGO). When components to which the TBC system has been applied stay at high temperatures longer, TGO layers composed of Al 2O 3 and other complex oxides grow thicker in the interfaces of the bond and top coats. 18) Thicker TGO layers lead to higher stress at the interface of the bond and top coats, leading to the formation and growth of cracks, coating layer delamination, and eventually damage to high-temperature gas turbine components. In this regard, the quality of the TGO layers is a critical factor that determines the lifetime of high-temperature components to which the TBC system is applied. Notably, TGO layers, which form and grow at the interface via an interaction between Al and oxygen, significantly depend on the chemical composition of the bond coat, and hence it is necessary to assess the effect of the bond coat composition on the formation and growth of TGO layers. 19-22)
The present study intends to determine how the chemical composition of the bond coat and the application of vacuum heat treatment affect the growth behavior of TGO layers and further the lifetime of the TBC system. To this end, TBC systems with two different types of bond coat were prepared, and were subjected or not subjected to vacuum heat treatment, which aimed to help relieve stress and improve interface stability. Subsequently, the effects of heat exposure time and vacuum heat treatment on the lifetime of the tested TBC systems were investigated. Among lifetime evaluation methods for the TBC system, the furnace cyclic test (FCT) was selected and employed to determine how the bond coat composition and vacuum heat treatment affected the growth behavior of the TGO layers.
2. Experimental Procedures
2.1. Starting Powder and Specimen Preparation
In the present study, a Ni-based super heat-resistant alloy (Nimonic 263, nominal composition of Ni-20Cr-20Co-5.9Mo-0.5Al-2.1Ti-0.4Mn-0.3Si-0.06C, in wt%, ThyssenKrupp VDM, Germany) was used as a substrate, and coin-shaped specimen with a diameter of 25 mm and thickness of 5 mm was prepared. Blasting treatment using Al 2O 3 powder was performed to facilitate coat formation, and the APS method was employed for the formation of both the top and bond coats. The bond coat was formed using two types of commercial feedstock powder as a starting material, as follows: AMDRY 962 (Oerlikon Metco Holding AG; average composition in wt%: Ni-22Cr-10Al-1.0Y; and average particle size: 53 - 106 μm) and AMDRY 386-4 (Oerlikon Metco Holding AG; average composition in wt%: Ni-22Co-17Cr-12Al-0.5Y-0.5Hf-0.4Si; and average particle size: 53 - 106 μm). The top coat was formed using METCO 204 C-NS (Oerlikon Metco Holding AG, Switzerland; 8YSZ; and average particle size: 45 - 147 μm). Subsequently, these coats were subjected to vacuum heat treatment for stress relief and interface stabilization, at 1,100°C for 3 h. Two types of heat-treated specimens and two types of non-heat-treated specimens were analyzed to determine the effects of the applied heat treatment and bond coat composition on the lifetime of the tested TBC systems. Schematic diagrams of the specimens used in this study are shown in Fig. 1.
2.2. Characteristics Evaluation
Cross-sectional microstructure of the heat-treated and non-heat-treated specimens were analyzed to appraise the occurrence of internal cracks in the coating layers and coat delamination, which might have occurred during the formation of the TBC system or vacuum heat treatment. The specimens were cut into pieces, mounted using epoxy resin, and polished using SiC polishing paper along with 1 and 3 μm diamond paste. Following the pretreatment, microstructure analysis was performed using a scanning electron micrography (JSM-5610, JEOL, Japan). FCT was employed to assess the effect of the vacuum heat treatment on growth behavior of TGO layers, while an X-ray Diffractometer (XRD, PANalytical, Model X’Pert PRO MPD) was used to observe phase transformation.
In general, the high-temperature components of gas turbines are exposed to repetitive heating and cooling during operation, which leads to the formation of the thermal and residual stresses at the interface of the bond and top coats, mainly ascribed to the thermal expansion mismatch between the top and bond coats of the system (8YSZ: 10.5-11.5×10 −6 K −1 and MCrAlY bond coat: 12.7-21.5×10 −6 K −1). 23,24) These stresses are, along with TGO layers, the main causes of interfacial delamination occurring at the bond and top coats, eventually shortening the lifetime of the TBC system. 25-27) In the present study, the FCT method, widely applied by advanced research institutes, was employed to assess the thermal durability of the TBC system. The test temperature was chosen to be 1,121°C, as in the advanced institutes. Specimens were heat treated for 25 h in an electric furnace and naturally cooled for 1 h at room temperature in a repetitive manner. 28) Heat exposure times were set to be 50, 100, 200, 400, and 1,000 h, and each test proceeded until the set exposure time, or until the occurrence of delamination. Schematic diagrams of the FCT equipment and test methods are shown in Fig. 2. Phase transformation in the top coat was observed and analyzed on the vacuum heat-treated specimens and non-heat-treated specimens before the FCT as well as specific specimens in which delamination occurred during the FCT.
3. Results and Discussion
3.1. Microstructure of TBC System
The microstructure of the vacuum heat-treated and non-heat-treated TBC specimens is shown in Fig. 3, and the thickness measurements of the bond and top coats are summarized in Table 1. Here, AMDRY 962 and AMDRY 386-4 were used for the bond coat feedstock for specimens (a) and (b), respectively, and their top coats were formed without the vacuum heat treatment. AMDRY 962 and AMDRY 386-4 were used for the bond coat feedstock for specimens (c) and (d), respectively, and their top coats were formed after vacuum heat treatment. The microstructure of the top coat, containing pores and oxides, is typical of the coating layer formed via the APS method, where splat interfaces arising from the cooling of melted powder particles can be observed. However, typical process defects (including horizontal or vertical cracks), which might form during the formation of coating layers, were not observed, and even after the heat treatment and pretreatment procedures, interfacial cracks or delamination were not observed. The bond coat layers contained oxides and some unmelted powders, but these are typically observed in bond coat layers produced via the APS method. In the meantime, any significant interfacial change at the top and bond coats was not observed, which arose from the applied vacuum heat treatment. Therefore, it was confirmed that a desired overall microstructure was achieved in accordance with the determined microstructure design plan.
3.2. Growth Behavior of TGO
Each TBC specimen was subjected to the FCT, and the heat exposure time was selected to be 50, 100, 200, 400, and 1,000 h; their cross-sectional micrographs are shown in Fig. 4. Here, TGO layers composed of α-Al 2O 3 are indicated as a solid line, while other mixed composition oxides of Cr 2O 3, NiAl 2O 4, and other oxides (spinel structure) are indicated by a dotted line. 29,30) During the initial 100 h, degradation of the TBC system was observed; black TGO layers composed of Al 2O 3 were formed and thickened at the interfaces of the top and bond coats. As the degradation time increased, Al depletion proceeded at the bond coat and Ni, Cr, and Co reacted with Al 2O 3 to form grey spinel structures. The formation of these spinel structures, which have a low fracture toughness, led to the early initiation of interfacial cracks in both the TGO and the top coat. Despite their stress-relieving effects to some extent, these cracks eventually deteriorate the mechanical properties of the interfaces, mainly causing coat delamination. 31,32)
The spinel structures were first observed at the interfaces of specimen (a), whose bond coat was composed of NiCrAlY (AMDRY 962) and which was not heat treated, after 200 h of the FCT. This observation partly explains the early occurrence of delamination in the top coat of specimen (a) during the FCT (after 875 h). In the case of specimen (d), which was heat-treated and whose bond coat was composed of NiCo-CrAlY-HfSi (AMDRY 386-4), delamination occurred in the top coat after 850 h. In both specimens, as shown in Fig. 4(a) and 4(d), delamination took place at the interfaces between the TGO layers and the top coats. In both specimen B (non-heat-treated, Fig. 4(b)) and specimen (c) (vacuum heat-treated, Fig. 4(c)), no delamination or cracks were observed until 1,000 h. These observations confirmed that the vacuum heat treatment improved the thermal durability of a TBC system whose bond coat is composed of NiCrAlY, while it deteriorated that of a TBC system whose bond coat is composed of NiCoCrAlY-HfSi. This way, the necessity of the vacuum heat treatment for each bond coat composition could be determined. The average thickness of the TGO layers in each specimen changes during the FCT is shown in Fig. 5, and those TGO thickness measurements at each FCT condition (heat exposure time) are summarized in Table 2. The average TGO layer thicknesses measured for specimens (a), (b), (c), and (d) after the initial 50 h were 2.5 ± 0.5, 2.6 ± 0.7, 3.4 ± 0.7, and 2.9 ± 0.5 μm, respectively. In all specimens, these TGO thickness measurements were at a similar level within the respective error range. After the initial 100 h, TGO layers thickened due to the thermal degradation in each specimen, i.e., 4.2 ± 0.7, 5.9 ± 1.8, 5.4 ± 0.9, and 3.5 ± 3.0 μm for specimens (a), (b), (c), and (d), respectively. It should be noted that the growth rate of the TGO layers was relatively low in specimen (d) while it was at a similar level in the other three specimens. After 200 h, TGO growth significantly accelerated in all specimens, i.e., 8.3 ± 3.6, 8.9 ± 3.0, 8.1 ± 1.5, and 6.0 ± 4.1 μm for specimens (a), (b), (c), and (d), respectively. After 400 h, the TGO layers, whose thickness exceeded 10 μm, the critical thickness, were observed in specific parts of all specimens except specimen (c) (indicated as a dotted ellipse in Fig. 5). Here, the average TGO thicknesses in specimens (a), (b), (c), and (d) were 12.3 ± 3.4, 10.4 ± 4.5, 9.8 ± 3.9, and 13.5 ± 6.0 μm, respectively. This means that, after 400 h, the TGO layer thickness in all specimens was close to, or larger than the critical thickness. Notably, the TGO thickness in specimen (d) measured after 400 h was much larger than that measured after 200 h. In specimens (a) and (d), delamination occurred after 875 and 850 h, respectively, and the TGO thicknesses were measured to be 20.3 ± 10.0 and 20.4 ± 11.1 μm, respectively. In specimens (b) and (c), where the FCT was carried out up to 1,000 h, the TGO thicknesses were measured to be at a level similar to those of specimens (a) and (d), i.e., 22.0 ± 5.2 and 19.3 ± 8.0 μm, respectively, but delamination did not occur. Based on these observations and analyses of the formation and growth behavior of TGO layers, it was confirmed that the TGO thickness exceeded the critical point when the TBC specimens were subjected to over 400 h of degradation. With regard to the formation and growth behavior of the TGO layers only, the NiCrAlY-based TBC system was superior to the NiCoCrAlY-based system for oxidation resistance due to its higher Ni content. The effect of the vacuum heat treatment on thermal durability, however, differed according to the bond coat composition. The NiCoCrAlY-based TBC system exhibited greater thermal durability without the vacuum heat treatment (as shown in specimen (b)), while the NiCrAlY-based system did so when heat-treated (as shown in specimen (c)). This phenomenon is attributed to the fact that, in the NiCrAlY-based system, the vacuum heat treatment promotes uniform distribution of β-NiAl, preventing the depletion of Al and therefore improving the system’s thermal durability. In the NiCoCrAlY-based system, however, the vacuum heat treatment helps Co enhance the resistance of coating layers against thermal stress and improve the system’s mechanical properties, including strength, but, at the same time, the treatment degrades the system’s oxygen resistance and thus thermal durability. In this regard, a future study will focus on establishing and verifying the optimized heat treatment conditions according to the bond coat composition (i.e., Ni and Co content), as well as appraising their effects on the system’s thermal durability.
3.3. Effect of Vacuum Heat Treatment and FCT on Phase Transformation in the Top Coat
To analyze the effects of the vacuum heat treatment and bond coat composition on the FCT lifetime, the top coat composed of yttria-stabilized zirconia (YSZ), where delamination occurred during the FCT, was observed and analyzed with respect to phase transformation. The focus of the analysis was to determine if any phase transformation had occurred, particularly from metastable tetragonal phases ( t′-YSZ) to tetragonal ( t-YSZ) or monoclinic ( m-YSZ) phases. 33) The corresponding results are shown in Fig. 6. In the top coat before the FCT ( Fig. 6(a)), the t′-YSZ phase, whose diffraction peak is located between 73° and 75°, was observed regardless of the application of the vacuum heat treatment, i.e. 1,100°C for 3 h, indicating that the phase transformation of the t′-YSZ phase into t-YSZ or cubic ( c-YSZ) phases did not occur. 33,34) In specimens (a) and (d), where delamination occurred after 850 and 875 h during the FCT, respectively, the t′-YSZ phase was partially transformed into the t-YSZ phase. The applied FCT conditions were considered to promote the transformation of these t-YSZ and/or c-YSZ phases into the m-YSZ phase. This transformation entailed 3 to 5% of volume expansion, causing delamination of the top coat in the TBC system. 14,18,35)
In this study, the effect of the bond coat composition on the phase transformation of the top coat, as well as the effect of the vacuum heat treatment on the microstructure evolution of the top coat were not significant. Also, the effect of the vacuum heat treatment on the phase transformation of the top coat was not determined. A future study will investigate in greater detail the relationship between the bond coat composition and the system’s phase transformation behavior in connection with degradation.
4. Conclusions
The present study aimed to determine the optimized bond coat composition and heat treatment conditions for thermal barrier coating (TBC) system by analyzing the effects of the bond coat composition and vacuum heat treatment on the growth behavior of thermally grown oxide (TGO) layers, as well as via phase transformation analyses.
The furnace cyclic test (FCT) results confirmed that the formation and growth behavior of TGO layers were affected by the bond coat composition and the application of the vacuum heat treatment. While the degradation time increased up to 400 h during the FCT, the TGO layer thickness increased in a linear manner, reaching critical thickness. The FCT was performed up to 1,000 h, and the results showed that the TBC system whose bond coat was composed of NiCrAlY exhibited greater thermal durability when vacuum-heat treated, while the NiCoCrAlY-based TBC system did so without the vacuum heat treatment. The top-coat layer of each TBC system was analyzed with respect to phase transformation. In vacuum heat-treated specimens, the transformation of metastable tetragonal phase (t′-YSZ) into tetragonal (t-YSZ) and/or cubic (c-YSZ) phases was not observed. In specimens where delamination occurred during the FCT, however, t′-YSZ phase was transformed into tetragonal t-YSZ phase. These observations explain the early initiation of delamination in specific TBC system under the applied FCT conditions.
Acknowledgments
This work was supported by the “Human Resources Program in Energy Technology” program of the Korea Institute of Energy Technology Evaluation and Planning (KETEP), granted financial resources from the Ministry of Trade, Industry & Energy, Republic of Korea. (No. 20174030201460) and by a National Research Foundation of Korea (NRF) grant funded by the Korean Government (MEST) (No. 2011-0030058).
Fig. 1
Schematic diagram for structural design of TBC as a function of heat treatment. 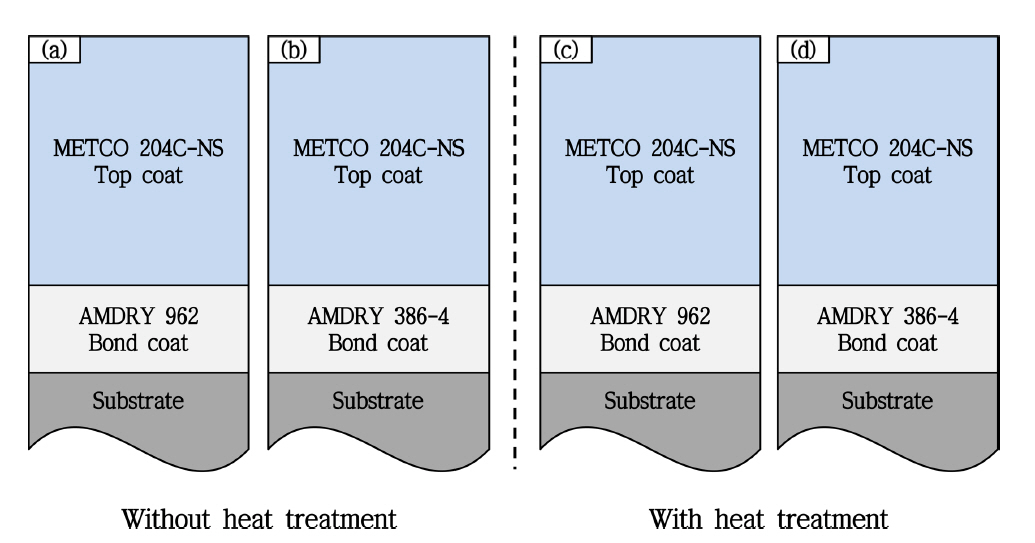
Fig. 2
Test apparatus and conditions applied in this study. 
Fig. 3
Cross-sectional microstructures of TBC specimens prepared with bond coat species and heat treatment: (a) TBC with 962 bond coat without heat treatment, (b) TBC with 386-4 bond coat without heat treatment, (c) TBC with 962 bond coat with heat treatment, and (d) TBC with 386-4 bond coat with heat treatment. 
Fig. 4
Interface microstructures after FCT for TBC specimens with bond coat species and FCT time: (a) TBC with 962 bond coat without heat treatment, (b) TBC with 386-4 bond coat without heat treatment, (c) TBC with 962 bond coat with heat treatment, and (d) TBC with 386-4 bond coat with heat treatment. 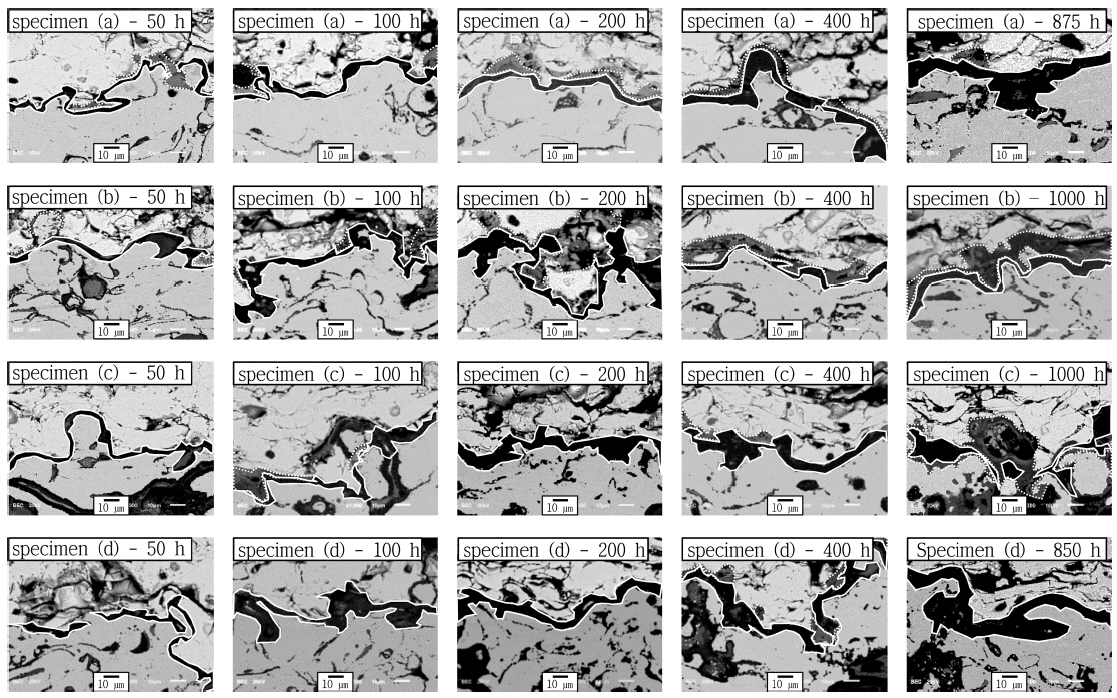
Fig. 5
Thickness of TGO layer for each TBC after FCT at 1121°C. 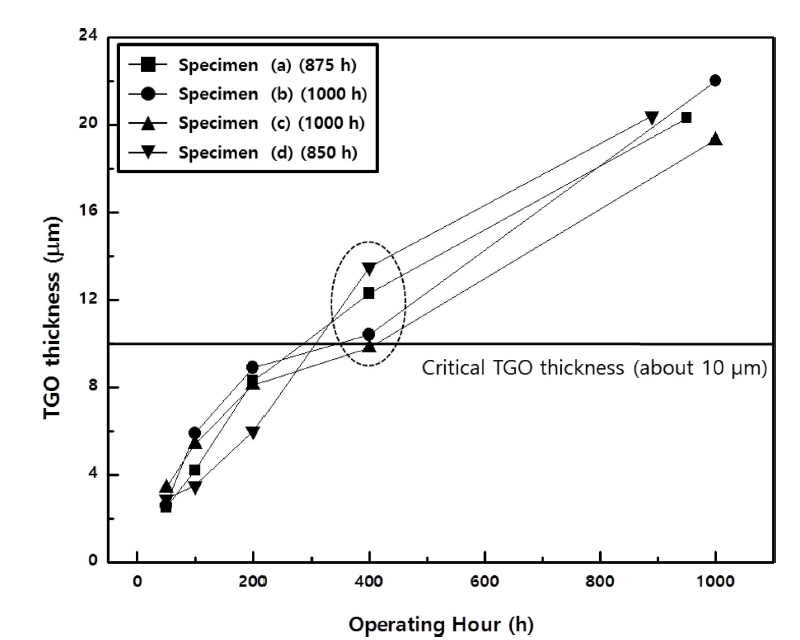
Fig. 6
Results of phase analysis for each top coat: (a) Before FCT with and without heat treatment and (b) After FCT with and without heat treatment. Red and blue peaks indicate specimens (a) and (b) of Table 1, respectively. 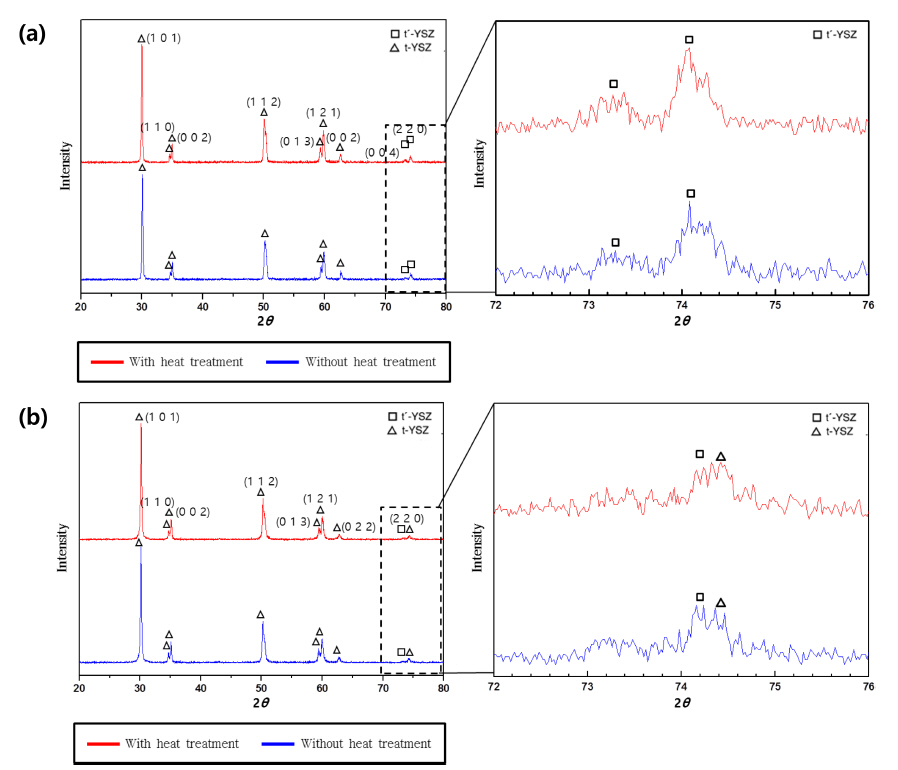
Table 1
Thicknesses of the Bond and Top Coats in TBC Systems Shown in Fig. 1
|
Specimen |
Top coat |
Bond coat |
|
Specimen (a) (AMDRY 962 bond coat, without heat treatment) |
794 ± 29 μm |
170 ± 21 μm |
|
Specimen (b) (AMDRY 386-4 bond coat, without heat treatment) |
831 ± 41 μm |
181 ± 17 μm |
|
Specimen (c) (AMDRY 962 bond coat, with heat treatment) |
805 ± 24 μm |
184 ± 37 μm |
|
Specimen (d) (AMDRY 386-4 bond coat, with heat treatment) |
906 ± 61 μm |
224 ± 12 μm |
Table 2
Thickness of TGO Layer for Each TBC System with FCT at 1121°C
|
Specimen |
Test hour in FCT |
Thickness of TGO layer |
|
Specimen (a) |
50 h |
2.5 ± 0.5 μm |
|
100 h |
4.2 ± 0.7 μm |
|
200 h |
8.3 ± 3.6 μm |
|
400 h |
12.3 ± 3.4 μm |
|
875 h |
20.3 ± 10.0 μm |
|
|
Specimen (b) |
50 h |
2.6 ± 0.7 μm |
|
100 h |
5.9 ± 1.8 μm |
|
200 h |
8.9 ± 3.0 μm |
|
400 h |
10.4 ± 4.5 μm |
|
1000 h |
22.0 ± 5.2 μm |
|
|
Specimen (c) |
50 h |
3.4 ± 0.7 μm |
|
100 h |
5.4 ± 0.9 μm |
|
200 h |
8.1 ± 1.5 μm |
|
400 h |
9.8 ± 3.9 μm |
|
1000 h |
19.3 ± 8.0 μm |
|
|
Specimen (d) |
50 h |
2.9 ± 0.5 μm |
|
100 h |
3.5 ± 3.0 μm |
|
200 h |
6.0 ± 4.1 μm |
|
400 h |
13.5 ± 6.0 μm |
|
850 h |
20.4 ± 11.1 μm |
REFERENCES
1. DR. Clarke, and SR. Phillpot, “Thermal Barrier Coating Materials,” Mater Today, 8 [6] 22-9 (2005).  2. R. Vaßen, MO. Jarligo, T. Steinke, DE. Mack, and D. Stöver, “Overview on Advanced Thermal Barrier Coatings,” Surf Coat Technol, 205 [4] 938-42 (2010).  3. M. Gell, E. Jordan, K. Vaidyanathan, K. McCarron, B. Barber, YH. Sohn, and VK. Tolpygo, “Bond Strength, Bond Stress and Spallation Mechanisms of Thermal Barrier Coatings,” Surf Coat Technol, 120-121 53-60 (1999).  4. KH. Stern, Metallurgical and Ceramic Coatings; pp. 194, Chapman and Hall, London, 1996.
5. SH. Jung, SH. Jeon, JH. Lee, YG. Jung, IS. Kim, and BG. Choi, “Effects of Composition, Structure Design, and Coating Thickness of Thermal Barrier Coatings on Thermal Barrier Performance,” J Korean Ceram Soc, 53 [6] 689-99 (2016).   6. H. Herman, S. Sampath, and R. McCune, “Thermal Spray: Current Status and Future Trends,” MRS Bull, 25 [7] 17-25 (2000).  7. U. Schulz, C. Leyens, K. Fritscher, M. Peters, SB. Bilge, O. Lavigne, JM. Dorvaux, M. Poulain, R. Mévrel, and M. Caliez, “Some Recent Trends in Research and Technology of Advanced Thermal Barrier Coatings,” Aero Sci Technol, 7 [1] 73-80 (2003).  8. TS. Sidhu, S. Prakash, and RD. Agrawal, “Studies on the Properties of High-Velocity Oxy-Fuel Thermal Spray Coatings for Higher Temperature Applications,” Surf Coat Technol, 41 [6] 805-23 (2009).  9. XQ. Cao, R. Vaßen, and D. Stöver, “Ceramic Materials for Thermal Barrier Coatings,” J Eur Ceram Soc, 24 [1] 1-10 (2004).  10. W. Schilke, Advanced Gas Turbine Materials and Coatings; GE Energy, Schenectady, New York, 1991.
11. T. Bhatia, A. Ozturk, L. Xie, EH. Jordan, BM. Cetegen, M. Gell, X. Ma, and NP. Padture, “Mechanisms of Ceramic Coating Deposition in Solution-Precursor Plasma Spray,” J Mater Res, 17 [9] 2363-72 (2002).  12. L. Xie, X. Ma, EH. Jordan, NP. Padture, TD. Xiao, and M. Gell, “Identification of Coating Deposition Mechanisms in the Solution-Precursor Plasma-Spray Process Using Model Spray Experiments,” Mater Sci Eng, A, 362 [1-2] 204-12 (2003).  13. KW. Schlichting, NP. Padture, EH. Jordan, and M. Gell, “Failure Modes in Plasma-Sprayed Thermal Barrier Coatings,” Mater Sci Eng, A, 342 [1-2] 120-30 (2003).  14. A. Rabiei, and AG. Evans, “Failure Mechanisms Associated with the Thermally Grown Oxide in Plasma-Sprayed Thermal Barrier Coatings,” Acta Mater, 48 [15] 3963-76 (2000).  15. NP. Padture, KW. Schlichting, T. Bhatia, A. Ozturk, B. Cetegen, EH. Jordan, M. Gell, S. Jiang, TD. Xiao, PR. Strutt, E. Garcia, P. Miranzo, and MI. Osendi, “Towards Durable Thermal Barrier Coatings with Novel Microstructures Deposited by Solution-Precursor Plasma Spray,” Acta Mater, 49 [12] 2251-57 (2001).  16. B. Barber, E. Jordan, M. Gell, and A. Geary, “Assessment of Damage Accumulation in Thermal Barrier Coatings using a Fluorescent Dye Infiltration Technique,” J Therm Spray Technol, 8 [1] 79-86 (1999).  17. RA. Miller, and CE. Lowell, “Failure Mechanisms of Thermal Barrier Coatings Exposed to Elevated Temperatures,” Thin Solid Films, 95 [3] 265-73 (1982).  18. WJ. Brindley, and RA. Miller, “Thermal Barrier Coating Life and Isothermal Oxidation of Low-Pressure Plasma-Sprayed Bond Coat Alloys,” Surf Coat Technol, 43-44 446-57 (1990).  19. E. Tzimas, H. Mullejans, SD. Peteves, J. Bressers, and W. Stamm, “Failure of Thermal Barrier Coating Systems under Cyclic Thermomechanical Loading,” Acta Mater, 48 [18-19] 4699-707 (2000).  20. AG. Evans, DR. Mumm, JW. Hutchinson, GH. Meier, and FS. Pettit, “Mechanisms Controlling the Durability of Thermal Barrier Coatings,” Prog Mater Sci, 46 [5] 505-53 (2001).  21. JC. Jang, and SC. Choi, “Numerical Simulation for Residual Stress Distributions of Thermal Barrier Coatings by High Temperature Creep in Thermally Grown Oxide,” J Korean Ceram Soc, 43 [8] 479-85 (2006).   22. JC. Jang, and SC. Choi, “Numerical Simulation of Effects of TGO Growth and Asperity Ratio on Residual Stress Distributions in TC-BC-TGO Interface Region for Thermal Barrier Coatings,” J Korean Ceram Soc, 43 [7] 415-20 (2006).   23. I. Taymaz, A. Mimaroglu, E. Avcı, V. Uçar, and M. Gur, “Comparison of Thermal Stresses Developed in Al2O3-SG, ZrO2-(12% Si+Al) and ZrO2-SG Thermal Barrier Coating Systems with NiAl, NiCrAlY and NiCoCrAlY Interlayer Materials Subjected to Thermal Loading,” Surf Coat Technol, 116-119 690-93 (1999).  24. JB. Wactman, “Mechanical Properties of Ceramics, an Introductory Survey,” Ceram Bull, 46 [8] 756-61 (1967).
25. E. Bakan, DE. Mack, G. Mauer, and R. Vaßen, “Gadolinium Zirconate/YSZ Thermal Barrier Coatings: Plasma Spraying, Microstructure, and Thermal Cycling Behavior,” J Am Ceram Soc, 97 [12] 4045-51 (2014).  26. R. Vassen, X. Cao, F. Tietz, D. Basu, and D. Stover, “Zirconates as New Materials for Thermal Barrier Coatings,” J Am Ceram Soc, 83 [8] 2023-28 (2000).  27. TA. Taylor, “Low Thermal Expansion Bondcoats for Thermal Barrier Coatings,” US Patent, 7,910,225 B2. March. 22. 2011.
28. A. Bolcavage, A. Feuerstein, J. Foster, and P. Moore, “Thermal Shock Testing of Thermal Barrier Coating/Bondcoat System,” J Mater Eng Perform, 13 [4] 389-97 (2004).  29. WR. Chen, X. Wu, BR. Marple, DR. Nagy, and PC. Patnaik, “TGO Growth Behavior in TBCs with APS and HVOF Bond Coats,” Surf Coat Technol, 202 [12] 2677-83 (2008).  30. O. Trunova, T. Beck, R. Herzog, RW. Steninbrech, and L. Singheiser, “Damage Mechanisms and Lifetime Behavior of Plasma Sprayed Thermal Barrier Coating Systems for Gas Turbines-Part I: Experiments,” Surf Coat Technol, 202 [20] 5027-32 (2008).  31. M. Daroonparvar, M. MazarAtabaki, M. Yajid, M. Sakhawathussain, M. Asgharifar, and NM. Yusof, “Microstrutural Characterization of Thermal Barrier Coating on Inconel 617 after High Temperature Oxidation,” Metall Mater Eng, 19 [2] 95-106 (2013).
32. DH. Lee, NK. Kang, KS. Lee, HS. Moon, HT. Kim, and C. Kim, “Evaluation of Thermal Durability of Thermal Barrier Coating and Change in Mechanical Behavior,” J Korean Ceram Soc, 54 [4] 314-22 (2017).   33. G. Witz, V. Shklover, W. Steurer, S. Bachegowda, and H. Peter Bossmann, “Phase Evolution in Yttria-Stabilized Zirconia Thermal Barrier Coatings Studied by Rietveld Refinement of X-Ray Powder Diffraction Patterns,” J Am Ceram Soc, 90 [9] 2935-40 (2007).  34. U. Schulz, and H-Tay. Lin, “Advanced Ceramic Coatings and Interfaces II,”; in A Collection of Papers Presented at the 31st International Conference on Advanced Ceramics and Composites, Wiley-InterScience, Florida, USA, 2007.  35. BC. Wu, E. Chang, SF. Chang, and CH. Chao, “Thermal Cyclic Response of Yttria-Stabilized Zirconia/CoNi-CrAlY Thermal Barrier Coatings,” Thin Solid Films, 172 [2] 185-96 (1989). 
|
|

















