1. Introduction
Photoelectrochemical (PEC) electrodes have often been applied to solve energy and environmental issues.1,2) To produce a PEC electrode, various catalysts can be considered candidates for practical applications. Semiconductor photocatalysts such as TiO2,3) Fe2O3,4) WO3,5) or ZnO6) have been studied as efficient materials in this field. Among those materials, TiO2 is one general material that has been explored as a semiconductor because of its non-toxicity, low cost, easy fabrication, and highly stable chemical and photo-response properties. However, TiO2 has the drawback of a large band gap (< 3.2 eV), and it absorbs only in the ultraviolet range, which is less than 5% of the solar spectrum. To reduce its band gap energy, TiO2 has been synthesized with various doping materials or heterogeneous structures including narrow band gap semiconductors such as ZnFe2O4,7) CuS,8) Cu2O9) or Bi2S3,10) etc.
Recently, polymeric materials of g-C3N4 (graphitic-carbon nitride) are being used as photocatalysts due to their lower band gap energy (> 2.6 eV) and excellent thermal stability at relatively high temperature.11-14) Particularly, g-C3N4 is a stable structure in acid or alkaline based solutions because of the strong covalent bonding of the C-N atoms.15) However, g-C3N4 exhibits fast recombination of electron and hole pairs, which leads to low efficiency in photocatalysis applications. To diminish the defects of g-C3N4, there have been several attempts to construct a unique composite. Hence, it has been reported that g-C3N4 possesses a higher conduction band than that of TiO2; this high conduction band is attributed to the couple g-C3N4/TiO2 composite as a stable heterojunction structure.16-19) In addition to this, to achieve better photocatalytic performance, 1D (1-dimensional) aligned TiO2 micro/nanorod structures have been explored for rapid electron transfer. However, these 1D structures have poor surface area and efficiency.20) Hence, to advance the overall performance, 3D (3-dimensional) constructions, such as branched or flower-like shapes, have been under profound consideration in attempts to increase the surface area and the photo-response efficiency. Such 3D structures can be more easily used to facilitate faster movement of electrons than is possible when using 1D structures.
In the present study, using hydrothermal and sintering methods, we explore the fabrication of a g-C3N4/TiO2 film. To control the band gap energy and to diminish the recombination rate as an effective method of separating electronhole pairs, g-C3N4 is coated on double layer TiO2 structures. Through the mechanism of its 3D nanostructure, g-C3N4/TiO2 can be used to demonstrate its potential. By loading a thin g-C3N4 layer on TiO2 double layer structures, electron scattering and absorption degree will increase more than is the case for a thick g-C3N4 layer because of the transmittance of incident light. Therefore, the use of a g-C3N4/TiO2 film as a 3D nanostructure facilitates electron transport under illumination; it is therefore possible to apply this method to PEC electrode technology.
2. Experimental Procedure
2.1. Preparation of TiO2 micro/nanorods
TiO2 micro/nanorods on an FTO substrate were prepared by following a basic hydrothermal process. Before employing the hydrothermal method, FTO substrates were cleaned with ethanol for 30 min. During the cleaning process, TiO2 solution was prepared. First, deionized water and HCl (Hydrogen chloride, Dae-jung) were mixed at a 1:1 volume ratio. Then, to prevent agglomeration of the Ti precursors, 1 mL of TIPP (Titanium isopropoxide, Sigma-Aldrich) was added drop-wise into the above solution and resulting mixture was stirred vigorously for 30 min. The two cleaned conductive FTO substrates were placed into a Teflon-lined autoclave facing the walls and the autoclave was heated to 150°C; this temperature was maintained for 3 h. After this, the autoclave was cooled to room temperature. Finally, the grown TiO2 film was rinsed with distilled water and dried at room temperature.
2.2. Sintering process for g-C3N4 on TiO2 film
The coating of the g-C3N4 layer was conducted in a crucible with a cover via a sintering process. For the formation of g-C3N4, a melamine precursor was obtained from Junsei Chemicals. Using this precursor, sample amounts of melamine if 0.25 g, 0.5 g, 0.75 g, and 1 g were added to the crucible and the prepared TiO2 film was placed facing down. The crucibles were then heated to 520°C and maintained at that temperature for 4 h to obtain uniform g-C3N4 layers; the heating rate was 10°min−1. Consequently, 0.25 g, 0.5 g, 0.75 g, and 1 g g-C3N4/TiO2 films were successfully obtained.
2.3. Photoelectrochemical performance
The photoelectrochemical (PEC) performance evaluation was conducted to measure the charge carriers under visible illumination (halogen lamp, 100 W). This process used a conventional three-electrode system. Carbon and Ag/AgCl were used for the counter and reference electrodes, respectively. 0.5 M Na2SO4 was used as the electrolyte. PEC was conducted with 1 V of applied bias potential versus the Ag/AgCl reference electrode during 4 cycles of on/off under visible light. To determine the long-term stability of the samples, a stability test was carried out for 5 h under the same conditions. LSV (Linear Sweep Voltammetry) analysis was carried out at a 20 mV/s scan rate and −1 to 1 V of potential range.
2.4. Characterization
XRD (X-Ray Diffraction; model D/Max-2500/PC, Rigaku, USA; Cu Kα source) measurement was performed to determine the crystalline structure properties of the TiO2 phase. The surface morphologies, used to differentiate between TiO2 and g-C3N4, were obtained by SEM (Scanning Electron Microscopy; model S4800, Hitachi, Japan) and TEM (Transmission Electron Microscopy; model JEM 2100F, JEOL, Japan). To observe and calculate the band gap energy of the films, optical absorbance measurement was conducted using UV spectroscopy (UV-Vis; model V650, JASCO, Japan); BaSO4 was used as the reference sample. FT-IR (Fourier-transform infrared spectroscopy; model iS10, Thermo Fisher Scientific, UK) measurement was conducted for observation of the chemical formation of g-C3N4. PL (Micro confocal Photoluminescence; model MonoRa750i, DongWoo Optron Co. Ltd, Korea) spectra measurement using a 325 nm excitation light source was conducted to determine the recombination rates of TiO2 and g-C3N4. PEC and LSV analyses were conducted using a potentiostat (model VersaSTAT4, Princeton Applied Research, USA)
3. Results and Discussion
3.1. Structural morphologies
3.1.1 SEM analysis
The surface morphologies of the distinctive g-C3N4 and TiO2 structures were observed using SEM. In Fig. 1(a), it can be seen that TiO2 micro/nanorods grew as double layer structures; these structures are present in rod shape in the bottom layer and in flower-like shape at the top of the layer. The overall length of the double layer was determined to have an average of 3 μm. Double layer structures are expected to accelerate the movement of electrons and to assist in improving the scattering effect. When the melamine powders were added to the TiO2 films to form the g-C3N4, the morphologies of the resulting mixtures revealed that g-C3N4 sheets were coated on the TiO2 surfaces according to the varying amounts of melamine of 0.25 g, 0.5 g, 0.75 g, and 1 g (Figs. 1(b) ~ (e)). From these microstructures, we can confirm that g-C3N4 was added to the TiO2 rod layer. Only 0.5 g and 1 g of g-C3N4/TiO2 were needed to uniformly cover the g-C3N4 on the TiO2 bottom layer. Moreover, 1 g of the g-C3N4/TiO2 structure was present as a film that was thicker than the other g-C3N4/TiO2 films. According to these results, we can confirm that g-C3N4/TiO2 structures were well-constructed with increasing of the added amounts of g-C3N4.
3.1.2 TEM analysis
To accurately determine the size of the g-C3N4 layer on TiO2 structure, TEM analysis was conducted, with results shown in Fig. 2. Here, we tried to compare only TiO2 rods and a 0.5 g g-C3N4/TiO2 film that we optimized as explained in later sections. As can be seen in Fig. 2(a), the average diameter was observed and found to be ~125 nm, which is in agreement with the SEM results. Importantly, coating thickness can be controlled according to the amount of added melamine. Figs. 2(b) and (c) shows that 0.5 g of g-C3N4/TiO2 leads to an approximately thin ~ 10 nm coating on the TiO2 rods. Further, according to the SAED (selected area electron diffraction) pattern, TiO2 and g-C3N4 showed crystalline and amorphous phases, respectively. The d-spacing of the TiO2 lattice is 0.34 nm (Fig. 2(d)). Therefore, we can confirm the crystallization of TiO2 from the lattice diffraction pattern and the g-C3N4 coating layers on the TiO2 rods.
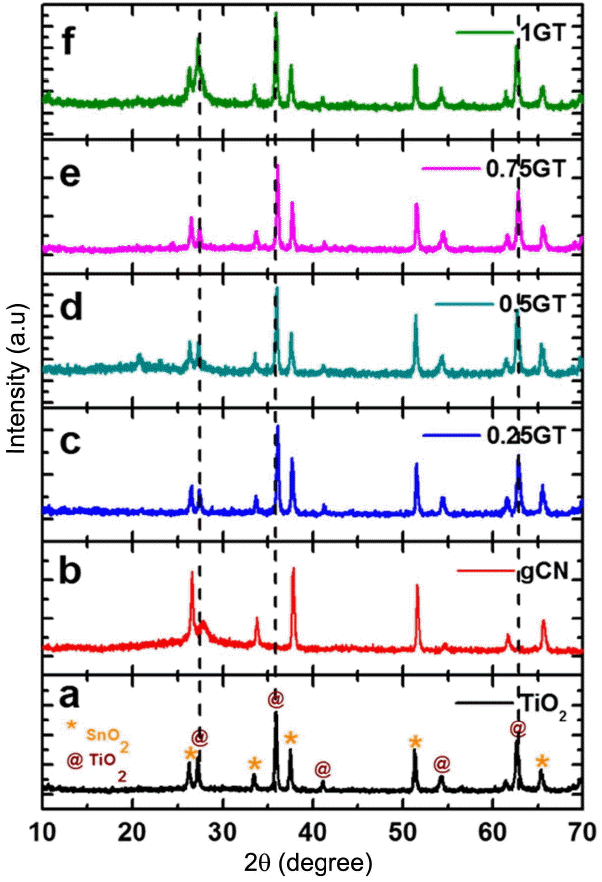
3.2. Phase crystallization - X-ray Diffraction
XRD analysis was conducted to examine the TiO2 crystallinity and the g-C3N4 phase structure. As can be seen in Fig. 3, the presence of only TiO2 peaks indicated the formation of rutile TiO2 phase, which is exactly correct according to JCPDS card No. 01-078-4190. Additional peaks in the TiO2 films can be referred to the SnO2 phase (JCPDS card No. 01-077-0449). Using XRD analysis, the TiO2 lattice constants were calculated and found to be a = 4.62, c = 2.15 Å and d110=3.26 Å for the tetragonal TiO2 structure. Pure g-C3N4 film showed a peak at around 27°, which means it has an amorphous structure as well, as can be seen in Fig. 3(b). However, it is difficult to differentiate TiO2 and g-C3N4 crystallization from Figs. 3(c) to (f) because the crystal (110) plane of TiO2 at 27° overlapped exactly with the g-C3N4 plane. The amounts of g-C3N4 can be correlated with the broadening of the peak at 27 compared to that of the pristine TiO2 film. Finally, it is proved that TiO2 is well-crystallized and that the g-C3N4 sheet is well formed with TiO2 micro/nanorods.
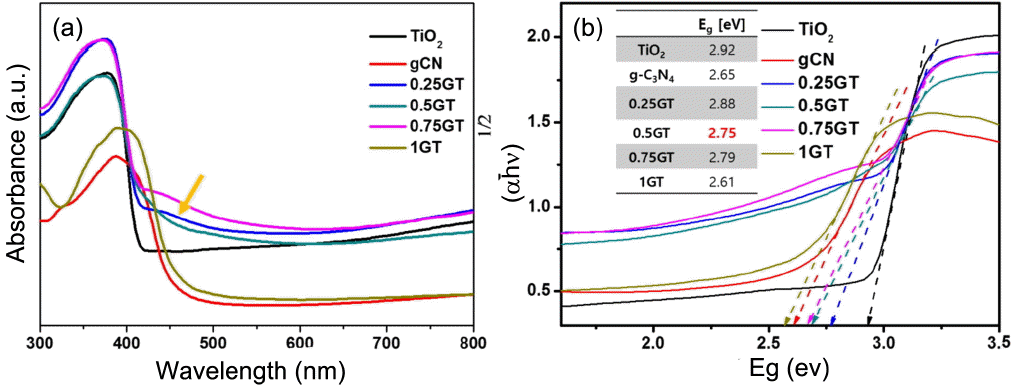
3.3. Optical absorbance
The degree of absorbance under the visible range can be determined using a UV-Vis spectrophotometer. All films were prepared for measurement of the optical absorbance from 800 to 300 nm. In Fig. 4(a), the absorbance properties of each film can be seen; the TiO2 film exhibited an absorbance edge at around 420 nm. The film of 0.5 g g-C3N4/TiO2 has an absorbance edge at 440 nm; this means that there is higher absorbance in the visible range. Pure g-C3N4 and 1 g g-C3N4/TiO2 film have similar slopes of around 450 nm. Fig. 4(b) presents detailed data on the calculation of the band gap energy of the films. The calculated band gap energies of the samples were 2.92, 2.65, 2.88, 2.75, 2.79, and 2.61 eV for TiO2, g-C3N4, 0.25 g g-C3N4/TiO2, 0.5 g g-C3N4/TiO2, 0.75 g g-C3N4/TiO2, and 1 g g-C3N4/TiO2, respectively. The band gap transitions are obviously revealed to depend on the amount of g-C3N4 added. Clearly, it can be seen that band gap energy can be adjusted according to the thickness of the g-C3N4 coating. From the band gap transition, because of the high optical absorbance, we can expect an effective generation of charge carriers.
3.4. FT-IR analysis
The existence of g-C3N4 was confirmed using the FT-IR spectrum (Fig. 5). The bands of g-C3N4 are mainly located from 1650 to 1200 cm−1. The band at 1625 cm−1 was assigned to a strong band of the C-N stretching modes. The other bands at 1401, 1317, and 1234 cm−1 were related to C-N bonding and represent heterocycle stretching of g-C3N4. The band at around 3000 cm−1 was found to correspond to H2O molecules adsorbed on the surface of the g-C3N4. The band at 805 cm−1 is indicated to the s-triazine ring vibrations of g-C3N4. However, 0.25 g g-C3N4/TiO2 and 0.75 g g-C3N4/TiO2 have no distinctive g-C3N4 peaks due to their insufficient coating on TiO2; the low degree of coating can be seen in the SEM images (Figs. 1(b)-(d)). Thus, the existence of g-C3N4 was directly confirmed through an observation of the chemical bonding.
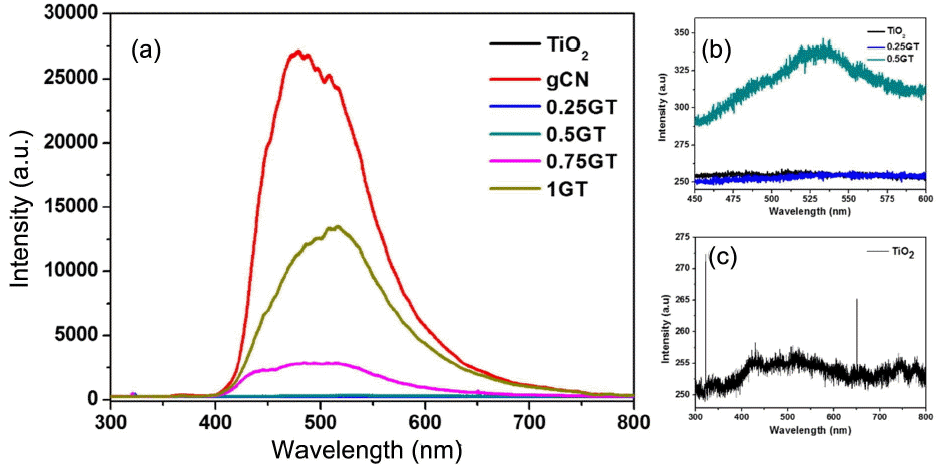
3.5. Photoluminescence
The possibility of charge separation due to g-C3N4 addition was observed using photoluminescence (PL) spectra, with results as shown in Fig. 6(a). The strength of PL intensity was found to increase according to the amount of added g-C3N4. This means that the recombination rate for this film is faster than that possible with any of the other films. From these results, pure g-C3N4 film was found to exhibit the highest intensity of around 500 nm. Then, the loadings of g-C3N4 were decreased from 1 g, to 0.75 g, and to 0.5 g of g-C3N4/TiO2. Fig. 6(b) shows that 0.5 g of g-C3N4/TiO2 film has the lowest intensity of PL due to the low recombination of charge carriers. Only the TiO2 and the 0.25 g g-C3N4/TiO2 films have almost no emission (Fig. 6(c)). The small PL intensity of the TiO2 film is the result of its relatively porous structure.21) On the other hand, due to the low level of loading of g-C3N4, the 0.25 g g-C3N4/TiO2 film showed trends similar to those of TiO2. Therefore, we identify that the 0.5 g g-C3N4/TiO2 film was better able to separate electron and hole pairs efficiently and to decrease the recombination rate than the 0.25 g, 0.75 g, and 1 g g-C3N4/TiO2 films.
3.6. Photoelectrochemical analysis
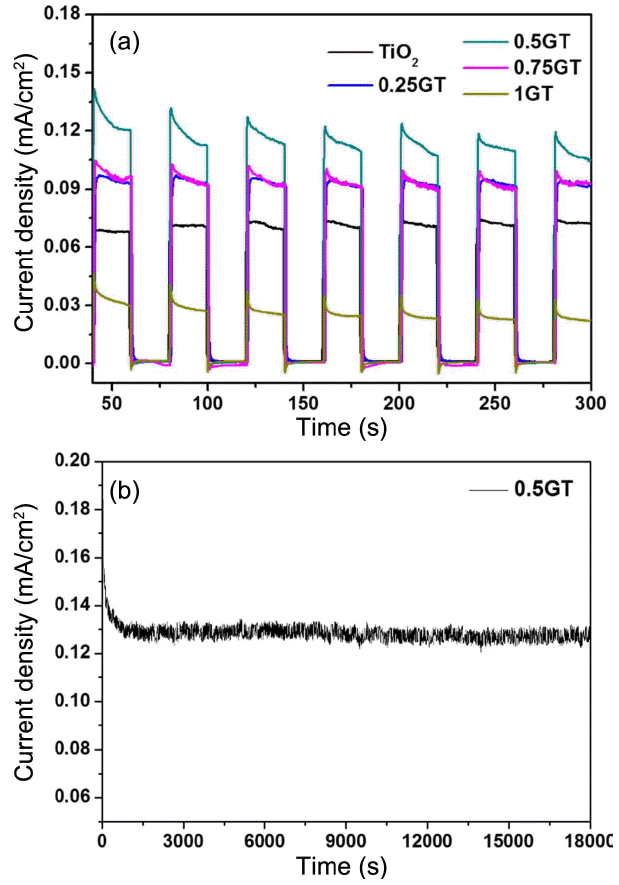
3.6.1 Photocurrent-time (I-t) curves
The evaluation of the PEC properties is a significant part of determining the chemical stability. The measurement of the current densities of the as-prepared films was conducted and results are shown in Fig. 7(a). Among the three films, the highest value of current density was obtained for the 0.5 g g-C3N4/TiO2 film at 0.12 mA/cm2. The second highest current density value was found in the 0.25 g and 0.75 g g-C3N4/TiO2 films, which both had a value of 0.1 mA/cm2. The TiO2 film and the 1 g g-C3N4/TiO2 film showed current density values of 0.75 and 0.03 mA/cm2, respectively. Interestingly, the thicker g-C3N4 film, namely the 1 g g-C3N4/TiO2 film, has the lowest current value, lower than even that of the TiO2 film. This trend could be seen to indicate that the addition of a larger amount g-C3N4 does not help the electron transfer efficiency. Moreover, a thicker coating layer might disturb the adsorption of light. Based on the PEC results, the 0.5 g g-C3N4/TiO2 film was used for confirmation of the photoresponse stability, as shown in Fig. 7(b). As well as a higher photoresponse under visible illumination, this film maintained a steady state of its original structure and did not decrease during the 5 h process. There needs to be a proper coating layer on the TiO2 micro/nanorods; 0.5 g g-C3N4/TiO2 seems to be a suitable material to enhance the PEC properties. Therefore, it is proved that an efficiently applied 0.5 g g-C3N4/TiO2 film can retain stability and can be used for PEC water splitting.
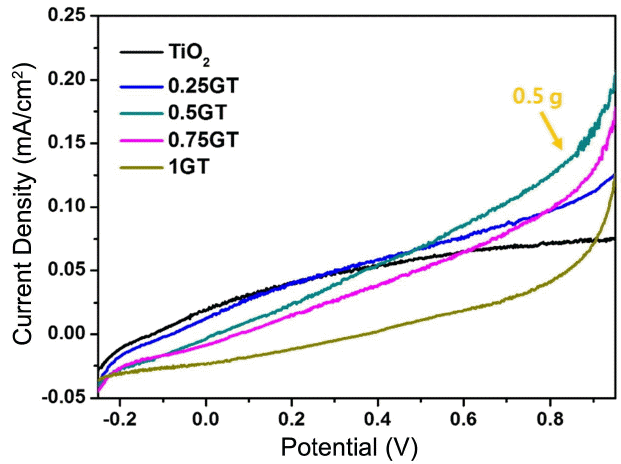
3.6.2 LSV
Figure 8 shows linear sweep voltammetry measurement results for TiO2 only and g-C3N4/TiO2 composite films under a visible lamp. The TiO2 film shows a 0.8 mA/cm2 current density, which is the lowest value among the samples. After loading the g-C3N4 on TiO2, a 1 g g-C3N4/TiO2 thick coating film leads to a current density value of 0.12 mA/cm2. Then, the thinner 0.5 g g-C3N4/TiO2 coating film has a value of 0.21 mA/cm2 for current density; this is approximately 3 times higher than the value of pristine TiO2 films (0.8 mA/cm2). The 0.25 g and 0.75 g g-C3N4/TiO2 films showed lower current density versus potential values than that of the 0.5 g g-C3N4/TiO2 film; however, this film did show a higher current density value than that of the 1 g g-C3N4/TiO2 film. Similar results for PEC and LSV also demonstrated that a thin coating of g-C3N4 can be used to enhance the current density. Once thick layers are coated on the TiO2 micro/nanorods, light was not able to reach the TiO2 surface. The electronic interaction between the TiO2 and the g-C3N4 layer can be used to separate the electrons and the holes efficiently, thus reducing the recombination rate (Scheme 1). Consequently, proper and uniform thickness of the coating layer is necessary for transmission of light to that of surface.
4. Conclusions
We successfully coated a g-C3N4 layer on 3D nanostructure grown TiO2 films via sintering method. Through micro-structural and morphological analyses, it was clearly seen that the 3D structure of TiO2 was grown as a double layer with a flower-like shape on aligned rods. Additionally, a g-C3N4 layer was coated onto the TiO2 double structure. From the FT-IR results, we confirmed the chemical bonding of g-C3N4 and the elements Ti, O, N, and C in the g-C3N4/TiO2 film. The g-C3N4/TiO2 films are used to extend the band gap into the visible range. Based on the optical absorbance property and PEC, the recombination rate for electron-hole pairs was found to have decreased. We can definitely prove that proper layer coating is necessary; thus, a 0.5 g g-C3N4/TiO2 film can lead to reduced recombination and enhanced electron transfer properties.