1. Introduction
Ce0.9Gd0.1O2-δ-La0.7Sr0.3MnO3±δ (GDC-LSM) ceramic is one of the most promising materials for oxygen separation among dual-phase ceramic oxygen transport membranes (OTMs), owing to its relatively high oxygen permeation flux and structural stability.1,2) From a theoretical point of view, OTM membranes should be as thin as possible to maximize the oxygen flux through the membranes.3,4) Therefore, a multilayered structure has been suggested, where the sealing/joining of ceramic membranes is essential. Moreover, the ceramic-to-metal joining is also needed because thin ceramic membranes need to be supported by metal supports with high joining strength. Therefore, the sealing/joining of OTMs with reliable microstructures, strengths, and in-service integrity has been the subject of substantial research over the last few years.5-7)
Reactive air brazing (RAB) is used widely to join ceramic-to-ceramic and ceramic-to-metal parts because of its simplicity, reliability, and cost effectiveness.8-16) The Ag-CuO system is considered a suitable brazing material for RAB because of its good wetting behavior on both ceramic and metal in air.7,12) Room temperature high strengths with a good balance between wettability and adhesion were observed by the addition of 1.4-8 mol% CuO in Ag matrix.9) Reliable joining and gas-tightness up to 800°C was reported using Ag-10 wt% CuO braze filler for joining GDC-LSCF ceramics with various metal alloys.7,15) The reliability of joints depends not only on the amount of Cu, brazing temperature, and time, but also on the thickness of the braze filler. Moreover, the bonding strength will be improved greatly if the joint is very thin.7,9,14) Nevertheless, realizing a desired thickness is extremely difficult when using conventional powder pressing or paste methods.
Many processes have been developed to prepare thin braze fillers, including screen printing, powder pressing, galvanization, electroplating, and thin foil formulation.12,17-21) Many of these processes require special equipment, which not only adds to the manufacturing cost but also extends the preparation time. Alternatively, researchers have developed a paste formulation to use as a braze filler, which requires organic additives, such as dispersant, binder, and solvent, to adjust the rheology.7,9) As the paste ages, however, its viscosity can change due to volatilization of the solvent, decreasing its flowability. In addition, the braze filler can be squeezed out if the amount of filler paste is high enough, suggesting that the realization of the uniform and thin joints is quite difficult using a paste.
On the other hand, tape casting is a well-known economic and feasible method for shaping thin ceramic tapes with a tailored thickness.22-24) Tape casting is used widely in the processing of multilayered ceramics, electro-ceramics, solid-oxide fuel cells, oxygen transport membranes, functionally-graded ceramics, structural materials, and many other applications.25-32) Indeed, tape casting of Ag with a thickness of 64±1 μm has also been reported.33) Tape cast interlayers have also been applied to many types of ceramic-to-ceramic and ceramic-to-metal joints.7,15,18,34,35) Therefore, this study examined the efficacy of tape casting of Ag-10 wt% CuO braze filler for the joining of GDC-LSM/SUS 460 FC by the RAB technique. The microstructures of the green tapes, interfacial microstructures and shear strengths of the asbrazed samples were examined.
2. Experimental Procedure
To prepare the Ag-10 wt% CuO paste, 3-roll milling was performed for the Ag paste (S-3902, Innovative Materials & Devices Inc., South Korea) after adding the appropriate amount of CuO powder (99.9%, Kanto Chemical, Japan), ethyl cellulose (Sigma Aldrich, USA) and α-terpineol (Sigma Aldrich, USA) to obtain a homogeneous Ag-CuO mixture. To convert this paste to a slurry for tape casting, a binder, plasticizer and solvent were added to the Ag-CuO paste followed by ball-milling for 24 h. Polyvinyl butyral (PVB) resin (Butvar B-98, Mw = 55,000 g/mol, Solutia, USA) was used as a binder, while a mixture of ethanol (40 wt%) and toluene (60 wt%) was used as the solvent. After dissolving 20 wt% of the binder in a toluene/ethanol mixture, 20 wt% of dioctyl phthalate with respect to the binder was added as a plasticizer, which imparted flexibility for the tape. Tape casting was performed on the slurry on a moving polyester film using a table top tape caster with a casting rate of 60 cm/minute. The cast tape was dried in air at room temperature. The resulting green tape thickness was approximately 120 μm. Ceramic pellets of 80 vol% Ce0.9Gd0.1O2-δ −20 vol% La0.7Sr0.3MnO3±δ (GDC-LSM) were consolidated to ≥ 98% by sintering at 1400°C. More details on the fabrication process are reported elsewhere.16) Thermal-resistant SUS 460 FC metal alloy (POSCO, South Korea), 2 mm in thickness, was chosen as the joining partner for the GDC-LSM ceramic. The SUS 460 FC was newly developed proprietary metal system for high temperature applications.
After cutting the GDC-LSM pellets and SUS 460 FC metal alloys, the surface for joining was polished to a 1 μm finish using diamond paste. The samples were cleaned ultrasonically in acetone and dried prior to joining. The filler tape was then inserted between the joining bodies; the same quantity of tape was applied to make a fair comparison. After binder burn-out at 400°C for 1 h, the samples were heat-treated in a box furnace at 1050°C for 30 min in air at a heating rate of 5°C/min and then cooled to room temperature. Three types of joint structures were prepared; a whole lap-joint for microstructural and elemental analyses, an offset lap-joint for the shear strength and butt-joint for flexural strength measurements.
The viscosity of the Ag-10 wt% CuO slurry was measured using a computer-controlled viscometer (LVDV-II+ Pro, Brookfield, USA) at 25°C. The green microstructures of the tape, joining interfaces and fractured surfaces after bending tests were characterized by scanning electron microscopy (SEM: S-4800, Hitachi). The shear strength of the joints was evaluated at room temperature using a universal testing machine (MTS Landmark, USA) at a head speed of 0.05 mm/min. For this test, the samples had an offset lap-joined structure based on ASTM D905.36) The shear test was performed on five samples under each condition to ensure the reliability of the joined samples. The flexural strength of joints was measured at RT, 500, 800 and 850°C using a three-point bending test. A minimum of three specimens with dimensions of 40 mm × 4 mm × 2.5 mm (length × width × thickness) were used for each measurement. The bending test was performed with universal testing machine (UTM) with a constant crosshead speed of 0.5 mm/min. For a high-temperature measurement, the specimens were dwelled at the testing temperatures in a box furnace for 10 min to reach thermal equilibrium prior to testing.
3. Results and Discussion
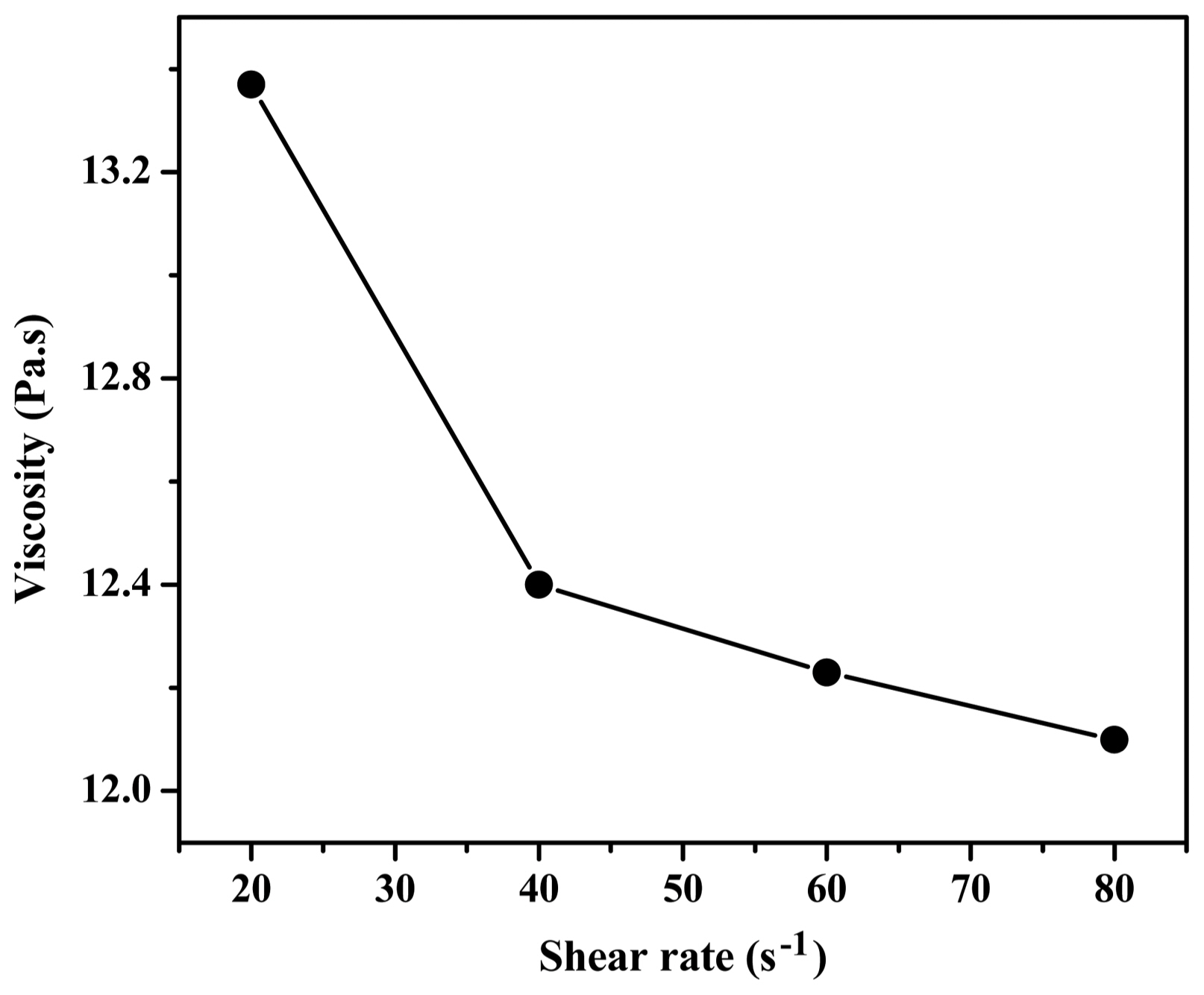
Shear thinning behavior and appropriate viscosity are essential for a slurry used for tape casting. A slurry with high viscosity cannot pass the blade, whereas uncontrolled flow and sedimentation occur when the viscosity is too low. For a slurry with an adequate rheology, the viscosity decreases when it passes the blade during the tape casting process due to pseudo-plasticity, and then it recovers after the slurry has left the blade by thixotropy. This shear thinning characteristic can avoid uncontrolled flow or sedimentation. 22,23) Fig. 1 shows the viscosity of the Ag-10 wt% CuO slurry. From the figure, it is evident that the viscosity decreases with increasing shear rate, which presents obvious shear thinning behavior or pseudo-plasticity of the slurry. The viscosity of the slurry was approximately 12 Pa·s at a shear rate of 40 s−1, which is satisfactory for tape casting.33)
Upon drying, the as-prepared green tape was highly flexible with no cracks, as shown in Fig. 2. Fig. 2 (b) shows the flexibility, where the dried cast tape was rolled around a pen without cracking and was unraveled easily without any residual deformation. Based on the flexibility of the tape, its handling should be easy when used as a filler material for brazing. The green tape had a uniform thickness of approximately 120 μm (Fig. 2 (d)), which is also beneficial for stacking. Fig. 3 presents the microstructures of the top and bottom surface of the Ag-10 wt% CuO green tapes. No macro-structural defects, such as bubbles, drapes or cracks, were observed, and the tapes were smooth without significant aggregation. The particle distribution was uniform at both surfaces. The bottom surface (Fig. 3 (b)) of the green tape has higher binder content than the top surface (Fig. 3 (a)) because the solvent volatilizes easily from the top surface compared to the bottom side during drying, which causes higher binder content at the bottom surface. The elemental mapping results revealed (not shown here) a uniform distribution of Ag and CuO in the tape. Therefore, tape casting is a convenient and reproducible way to produce a homogeneous, thin, and flexible Ag-10 wt% CuO tape for RAB.
Figure 4 gives an overview of the cross-sectional SEM image of typical GDC-LSM/SUS 460 FC joint. The braze filler wetted both the ceramic and metal alloy sides well, and the interfaces were sound and crack-free with good physical contact. The observed microstructure was similar to other ceramic-to-metal joints brazed using Ag-10 wt% CuO paste.7,16) CuO did not react with the ceramic part to form an intermediate compound; it tended to precipitate and nucleate at the interface during cooling in the Ag matrix.15) Four different regions were observed in the SEM images and labeled as C (ceramic), F (braze filler), O (oxidation layer), and M (metal alloy). The brazed region (F and O) had a thickness of approximately 75 μm. The middle area of the brazed region, F, was an Ag-rich region with a small amount of precipitated CuO. The O (oxide) zone adjacent to the SUS 460 FC at the filler/metal alloy interface has a mean width of approximately 25 μm, which was formed by the oxidation of various metal elements diffused from alloy. The oxides precipitated in the Ag matrix. According to EDS analysis data for oxide layer (Fig. 4 (b)), shown in Table 1, the oxide layer is composed mainly of Fe, Cr, O and Cu elements. The composition of the oxide layer consisted of CuO, FexOy, and CrxOy. This oxide layer composition is consistent with the literature data of similar SUS metal alloys,7,16) which revealed the formation of Cr2O3, Fe2O3, Fe3O4, and FeCr2O4 phases by the oxidation of metal alloys in air. Therefore, this oxide layer was formed by the oxidation of Fe and Cr that diffused out from SUS 460 FC and the oxidation of Cu from the braze filler. According to the calculated Gibbs free energy,16) the formation of oxides from the metallic constituents of SUS 460 FC near 1050°C is inevitable when RAB is performed in air.
The mean shear strength of the as-brazed joints was 60±8 MPa at room temperature, which is comparable to other similar ceramic-to-metal joints.7,16) Fracturing occurred mainly in the GDC-LSM ceramics or barely at the joining interfaces (cohesive failure) upon the shear strength test. Fig. 5 shows a corresponding fractographic SEM image of a typical joint after cohesive failure. A uniformly distributed and densely packed Ag-10 wt% CuO braze filler was observed on both surfaces of ceramic and metal alloy. Fracturing at the GDC-LSM ceramics rather than the joining interface indicates that the joining part is mechanically stronger than the GDC-LSM ceramic. Flexural strength data of joints are shown in Fig. 6 as a function of temperatures up to 850°C. The strengths at RT and 500°C of the joints were 78±6 MPa and 75±8 MPa, respectively. Strength retention at 500°C was almost 100% for the joints. However, the joints showed a clear degradation in strengths at 800 and 850°C. The strength retentions at 800 and 850°C were 57% (45±7 MPa) and 37% (29±7 MPa) respectively. The strength degradation from 78±6 MPa at RT to 29±7 MPa at 850°C can be attributed to the softening nature of Ag-CuO filler at high temperatures. Overall, the powder or paste form filler can be replaced with a thin tape for reactive air brazing.
4. Conclusions
The efficacy of tape casting was examined as a potential replacement for the commonly used Ag-CuO powder or paste-form filler for reactive air brazing. The Ag-10 wt% CuO braze tape filler revealed successful joining properties for the sintered Ce0.9Gd0.1O2-δ -La0.7Sr0.3MnO3±δ (GDC-LSM) and SUS 460 FC metal alloy joint for oxygen transport membrane applications. The as-prepared green tape was quite flexible with no cracks, and could be used in any shape after cutting with easy handling. Green tape with a smoothness and a homogeneous distribution of Ag and CuO particles without macro-structural defects was prepared with a thickness of 120 μm. After heat treatment at 1050°C for 30 minutes in air, a reliable and compact GDC-LSM/SUS 460 FC joint was obtained with an average shear strength of 60±8 MPa. High temperature flexural strength of joints revealed strength retention of almost 100% at 500°C. However, the strengths were degraded to 57% and 37%, respectively, at 800 and 850°C.