1. Introduction
In residential buildings, humidity control and ventilation are factors critical for human heath as well as for the energy efficiency and durability of buildings. Thus, there is an urgent requirement for developing interior and exterior building finish materials, which can control humidity and ventilation in buildings. A humidity control ceramic is a functional material that controls the humidity of a room by the condensation or evaporation of water, which occurs at a specific humidity level, according to Kelvin’s capillary condensation theory.1) As this ceramic contains uniformly sized mesopores, it condenses or liquefies vapor when humidity increases while simultaneously evaporates at low humidity so as to control indoor humidity within a range of 40% - 70%.2) Besides controlling the indoor humidity, the humidity control ceramic effectively absorbs or eliminates environmentally hazardous material, such as volatile organic compounds (VOCs), which causes sick house syndrome or atopic dermatitis, as well as provides insulation effects; hence, it has attracted attention as an environment-friendly building material that could substitute cement or concrete material.3)
On the other hand, the performance of a humidity control ceramic is determined by its water adsorption/desorption capability and rate, and for attaining superior performance, it should have uniformly sized mesopores ranging from a few nanometers to 10 nm and macropores with a size of greater than or equal to 30 nm, which constrain the diffusion of water vapor.4) Typically, lime mortar, humed silica, silica gel, and zeolite cement are known to be humidity control materials; however, these materials exhibit low strength and durability and are not cost-effective.5-8) Recently, super porous minerals with a large specific surface area and meso-sized pores, such as diatomites, volcanic ash, and sepiolite, have attracted attention as humidity control ceramic materials.9-11)
Drinking-water treatment sludge (DWTS) is generated by the elimination of impurities from raw water by precipitation, filtration, and chemical treatment at purification plants; it is disposed of by dumping in sea or landfill. Recently, dumping DWTS into the sea has been restricted ever since the London Convention came into effect; moreover, the cost of landfills has been increasingly steadily. DWTS mainly consisting of silica (SiO2) and alumina (Al2O3) contains cohesive agents containing Fe+3 or Al+3 and small amounts of organic matter; as compared to sewage sludge, it is less environmentally hazardous, and it is rather similar to clay soil, which is more favorable for recycling into building material.12,13) Onggi soil is a natural mineral, which is used to produce earthenware; it is resistant to acids and alkali, and it exhibits low thermal conductivity and a high insulation effect when fired at appropriate temperatures. Furthermore, it exhibits high fluid tightness and strength as it has very fine pores. Thus, it is called “breathing pottery,” with increased utilization.14)
In this study, environment-friendly humidity control ceramics, which exhibit superior water adsorption and desorption capabilities, were fabricated from DWTS and Onggi soil. DWTS and Onggi soil mixed at different weight ratios were fired at 800 - 1000°C. Densification behavior and microstructure were investigated in terms of the firing temperature and the DWTS to Onggi soil weight ratio. The water adsorption and desorption capabilities of the sample were evaluated for determining the applicability as a humidity control ceramic.
2. Experimental Procedure
First, DWTS powder was prepared by drying and heat-treating dewatered sludge cake, which was supplied from the Incheon Bupyeong drinking water purification plant, at 80°C and 700°C, respectively. Weight loss attributed to the evaporation of water and the decomposition of residual water and organic matter was estimated to be 70% - 80% and 35%, respectively. After drying and heat-treatment, the obtained DWTS powder was highly agglomerated; thus, it was pulverized for 12 h using a planetary mill (Pulverisette 6, Fritsch) using a 2 mm yttria-stabilized zirconia (YSZ) ball and distilled water.
Onggi soil was purchased from Suksan Ceramics, Korea. As the Onggi soil used herein partly contains large silica particles, it was sieved using a 150 mesh (≤104 μm). First, the DWTS powder and Onggi soil were mixed using a planetary mill for 12 h. The weight ratios of DWTS:Onggi soil were 40:60, 50:50, 60:40, and 70:30. Second, the powder mixture was fired in an electric furnace at 800-1000°C. Given the potential damage to the sample by the gas generated during the decomposition of residual organic matter in DWTS powder, the heating rate was limited to 2°C/min for allowing the gas to leak out in the process.
The composition of the DWTS powder and Onggi soil was analyzed by ICP-OES (Optima 5300 DV, Perkin Elmer). The specific surface areas of the dried and fired powder samples were measured by N2 adsorption and desorption (TriStar II 3020, Micromeritics). The powder was pretreated at 350°C for 6 h. The density and porosity of the samples were measured by the Archimedes method. The microstructure was monitored by field-emission scanning electron microscopy (FE-SEM, S-4300, Hitachi).
A bar specimen with dimensions of 3 × 4 × 40 mm, according to KS (KS L 1591), was fabricated for measuring the flexural strength of the sample. All bar specimens were fractured in the four-point bending mode using a universal testing machine (UTM). The fracture load test was conducted using a jig with an external span of 30 mm and an internal span of 10 mm. The cross-head speed was 0.5 mm/min. The flexural strength s was calculated by the following equation.
Here, P is the fracture load (N); L and l are lengths of the outer and inner spans, respectively; b and d are the width and thickness of the sample, respectively.
The adsorption and desorption capabilities of the samples were measured using a thermo-hygrostat (TH-I-180, Jeio Tech). The samples were pretreated at a temperature of 25°C and a humidity of 50% for 24 h. Adsorption by the pretreated sample was conducted at a temperature of 25°C and a humidity of 95% for 12 h, and adsorption capability was evaluated by measuring the weight. For desorption, the sample after the adsorption test was left at a temperature of 25°C and a humidity of 30% for 12 h before measuring the weight to check the desorption capability. For minimizing error, these adsorption and desorption tests was repeated four times under the same test conditions.
3. Results and Discussion
Table 1 summarizes the results obtained from ICP analysis of the DWTS powder and Onggi soil; these materials are mainly composed of SiO2, Al2O3, and Fe2O3. The content of Al2O3 in DWTS was greater than that in Onggi soil, attributed to aluminates used as a cohesive agent in purification plants. The higher silica content in Onggi soil as compared with that in DWTS is attributed to silica and clay.
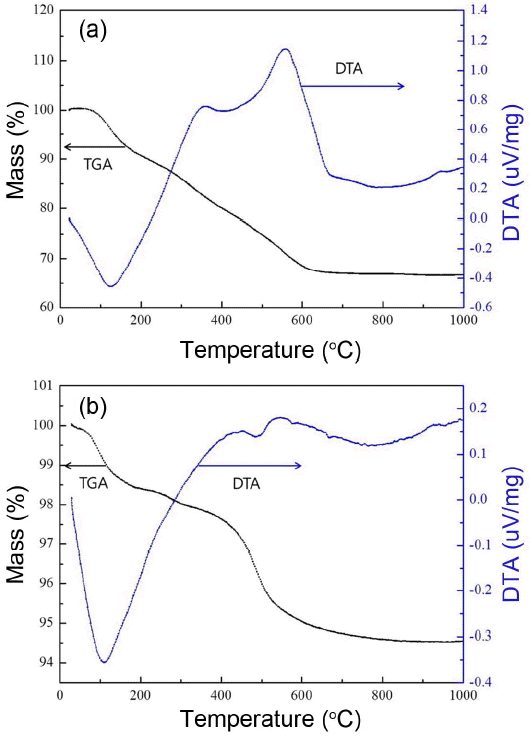
Figure 1 shows the TG-DTA curves of DWTS powder and Onggi soil. Exothermic peaks were observed at 100°C for both samples, attributed to the evaporation of adsorbed moisture. In Fig. 1(a), large exothermic peaks were observed at 360° and 580°C in DWTS, attributed to the organic matter generated from the purification and decomposition of the cohesive agent, and a modest endothermic peak was observed at 400 - 500°C. During heat-treatment, odor was generated from the decomposition of organic matter till 360°C, which was then eliminated with increasing temperature. Given this fact, the exothermic peak at 360°C was attributed to the decomposition of organic matter, while the exothermic peak observed at 580°C was attributed to the decomposition of the cohesive agent. In addition, the endothermic peak observed between 400 and 500°C was attributed to the evaporation of the water of crystallization from clay minerals in two powders. In Fig. 1(b), a endothermic peak, as well as significant weight loss, was observed at 410 - 600°C, attributed to the generation of metakaolin (Al2-Si2O7) coupled with the dehydration of kaolinite (Al2-Si2O5(OH)4) in Onggi soil.
From the results obtained from the TG-DTA analysis of DWTS and Onggi soil, the evaporation of the water of crystallization, decomposition of organic matter, or phase changes were not observed at a temperature of greater than or equal to 800°C. Given this result, it is imperative to set the firing temperature at a temperature of greater than or equal to 800°C for the manufacturing of a humidity control ceramic from DWTS powder and Onggi soil. Moreover, with increasing firing temperature to greater or equal to 1000°C, pores in DWTS powder and Onggi soil were rapidly eliminated, possibly resulting in the loss of function of the humidity control ceramic. Hence, for fabricating the humidity control ceramic from DWTS powder and Onggi soil, the firing temperature was set at 800-1000°C.
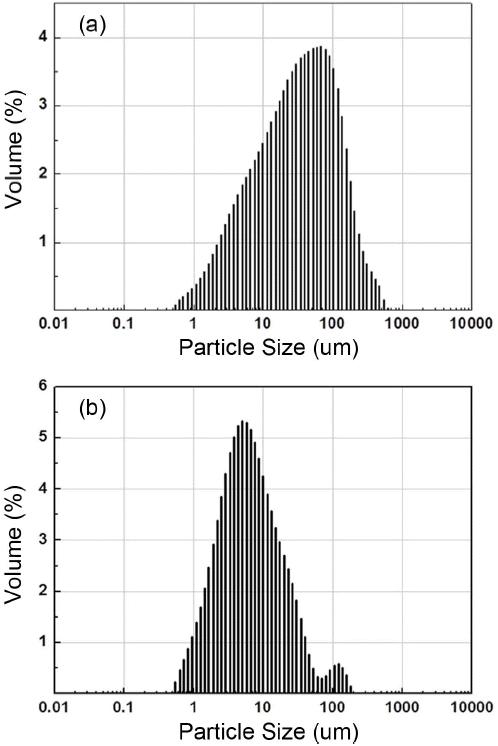
Figure 2 shows the results obtained from the particle size analysis of DWTS powder and Onggi soil, pulverized for 12 h after heat treatment at 700°C. As DWTS is a product obtained from the precipitation or cohesion of a water purification plant, its particle size distribution ranges from a few micrometers to a few tens of micrometers. On the other hand, as compared to DWTS powder, Onggi soil is refined material, with a more fine and narrow grain size distribution, ranging from 3 to 4 μm.
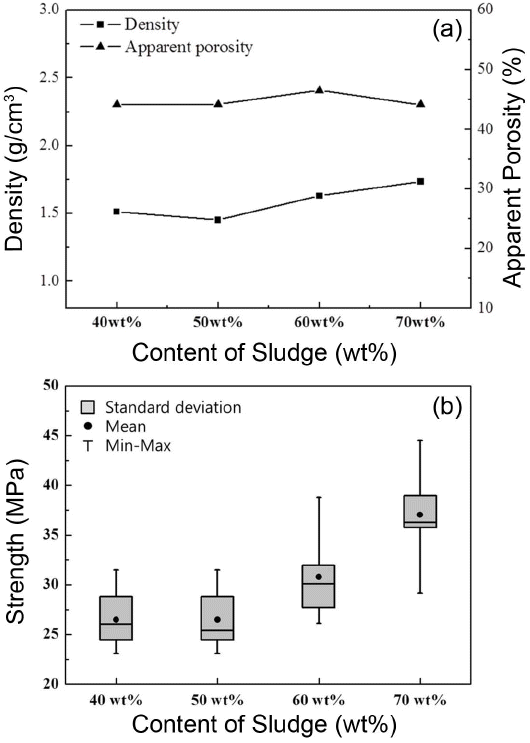
Figure 3 shows the density, porosity, and bending strength of the sample fired at 985°C. The weight ratios of DWTS:Onggi soil samples were 40 : 60, 50 : 50, 60 : 40, and 70 : 30. As shown in Fig. 3(a), with increasing DWTS content, density slightly increased, while porosity remained almost constant. The density and porosity of the sample containing 70 wt% of DWTS were 1.73 g/cm3 and 44%, respectively. As compared to Onggi soil, DWTS powder contains more alkali cations, such as CaO, Na2O, and K2O; thus, it tends to form eutectic mixtures, which form liquids at low temperature by reaction with silica and alumina during firing. As a result, with increasing DWTS content, density increases accordingly because of accelerated sintering.15,16) With increasing DWTS content, density also increased, albeit porosity remained constant, rather of drastic reduction, attributed to the residual organic matter present in DWTS, which served as a pore former.
As shown in Fig. 3(b), with increasing DWTS content, bending strength improved. Given that density increases with dependence on DWTS content and that bending strength is sensitive to sample density, the results observed in Fig. 3(a) and (b) are in agreement. For instance, the density of samples with 40% and 50 wt% of DWTS was insignificant, as well the strength remained constant, implying a close relationship between density and strength. Moreover, besides improvement in density, the bending strength of 60% and 70 wt% DWTS samples also significantly improved, attributed to another reason. As mentioned before, the eutectic mixture is expected to increase with increasing DWTS content, and for a sample containing a large DWTS content, bending strength is expected to significantly improve because of improvement in necking among the particles.
Table 2 summarizes the density, porosity, and specific surface area of samples fired at various temperatures. The ratio of DWTS:Onggi soil was 70 : 30. Density increased with firing temperature, while porosity decreased. Then, the specific surface area significantly decreased with increased firing temperature. For instance, the porosity and specific surface area of the sample fired at 800°C was 51.0% and 42.5 m2/g, respectively, and porosity decreased to 45.3% at a firing temperature of 900°C, although specific surface area decreased to the half or more. Typically, densification gradually progresses with increasing firing temperature, resulting in increased density and grain growth; thus, specific surface area significantly decreases. Particularly, with increasing firing temperature to 800 - 900°C, as compared to densification, particle growth increased. Thus, it is necessary to fire samples at temperatures of less than or equal to 900°C for producing a sample with superior humidity control performance.
Figure 4 shows the SEM images of the fracture surfaces of the samples fired at 800, 900, and 985°C from the sample with a DWTS:Onggi soil weight ratio of 70 : 30. No significant difference in porosity was observed because of the increased firing temperature; however, particle size changed. With increasing firing temperature from 800 to 900°C, particle size also significantly increased, and in particular, particle growth was considerable, and necking and a dense microstructure were observed in the sample fired at 985°C. Moreover, with increasing temperature, a network structure consisting of fine particles was formed, and nano-sized pores were eliminated first, affording a dense microstructure. As described in Table 2, specific surface area rapidly decreased, which was attributed to densification and particle growth with increasing temperature.
Figure 5 shows the water adsorption and desorption capabilities of the samples fired at 800, 850, 900, 975, and 985°C. The weight ratio of DWTS:Onggi soil was 70 : 30. As can be expected from the specific surface areas listed in Table 2, the lower the firing temperature, the higher the adsorption and desorption capabilities, and the capability of the samples fired at temperatures of greater than or equal to 900°C significantly decreased, attributed to the elimination of mesopores as well as rapid particle growth caused by firing temperature.
In this study, the fabricated humidity control ceramics exhibited a bending strength of 25.8 MPa, a specific surface area of 42.5 m2/g, and adsorption and desorption capabilities of 439 g/m2 and 422 g/m2, respectively, while the bending strength, specific surface area, and adsorption and desorption capability of a commercially available humidity control tile were 22.9 MPa, 37.4 m2/g, and 400 g/m2, respectively.17) Hence, DWTS and Onggi soil demonstrate superior performance and can be employed as humidity control ceramic materials.
4. Conclusions
In this study, an humidity control ceramic with superior adsorption and desorption capabilities was produced using DWTS and Onggi soil. Although the density slightly increased with increasing DWTS content, porosity decrease was insignificant, while specific surface area increased, indicating that the performance of humidity control ceramic increases with increasing DWTS content. For the sample with a DWTS : Onggi soil weight ratio of 70 : 30, porosity and specific surface area decreased with increasing firing temperature, as well as adsorption/desorption capability. At a firing temperature of greater than or equal to 900°C, the porosity decrease for the samples was insignificant, although specific surface area and adsorption/desorption capability significantly decreased because of rapid particle growth. The specific surface area and adsorption/desorption capability of the sample with a DWTS:Onggi soil weight ratio of 70 : 30 fired at 800°C were 42.5 m2/g and 439 g/m2 and 422 g/m2, respectively, demonstrating superior adsorption and desorption performance.