1. Introduction
Porous materials, a term referring to materials having a porosity from 15% to 95%, have unique characteristics that dense materials do not have, including thermal blocking, separation, and storage. In particular, porous ceramics are used in various areas, such as membranes, water treatment filter, thermal insulators, and sensors, and their applications are gradually extending.1-4) Porous alumina, one of the porous ceramics, is in good supply because the price is relatively low, and the material may be manufactured by mass production because it may be sintered in general atmosphere. In addition, due to its excellent mechanical, physical, chemical, and thermal properties, porous alumina is widely used in industry, and applied to the parts of manufacturing devices in the semiconductor and display industries. Despite these advantages, because porous alumina has a high electric resistance of about 1 × 1015 Ω·cm, its application to the parts of semiconductor manufacturing devices is limited as anti-static treatment is required. A vacuum chuck, a transportation device used in semiconductor processes, may be used as a work stage for loading a wafer in a semiconductor manufacturing device. Among vacuum chucks, the vacuum chuck for flat panel display (FPD) is a device that fixes a substrate by vacuum adhesion. Substrates for FPD, having a high electric resistance near to that of an insulator, form static polarization when in contact with the vacuum chuck, which also has a high electric resistance. Spark discharge caused by the static polarization may result in damage to the substrate.5) Therefore, the application of alumina to vacuum chucks used in semiconductor processes requires the control of the electric resistance of alumina, which requires further research. Methods of controlling the electric resistance of alumina include addition of a metal oxide to alumina: TiO2 may be added to alumina to produce excess electrons.6) In addition, Mn-based oxide may be added to form a low electric resistance secondary phase, such as Mn-Al spinel, to decrease the electric resistance of alumina.7) A vacuum chuck is a device that adheres to a substrate for transportation through vacuum suction. Therefore, a vacuum chuck requires excellent permeability to achieve high vacuum suction force. Efforts have been made to prepare porous alumina having a high permeability by using coarse alumina powder to form large open pores between powder particles.
In the present study, an experiment was performed to prepare a porous alumina material applicable to a vacuum chuck for semiconductor or display processes; the effects of MnO2 content on the strength, permeability, and electric resistance were investigated.
2. Experimental Procedure
In the experiment, to investigate the properties depending on the MnO2 content, specimens were prepared using coarse alumina powder. As shown in Table 1, the alumina powder used in the experiment was Al2O3 #280 Mesh (~ 33.43 μm, 99.51% white aluminium oxide grit, Kramer Industries, USA). To control the electrical properties, metal oxides were added. TiO2 (99.5% Sigma-Aldrich, U.S.A.) was added at 2 wt%, and MnO2 (99% Sigma-Aldrich, U.S.A.) was added at 2, 4, 6, 8, and 10 wt%. Table 2 shows the compositions of the mixtures. After weighing the mixture powder, dry ball-milling was performed for 12 h. Polyethylene glycol (PEG, Sigma-Aldrich, U.S.A.) was added as a plasticizer; then, the mixture powder was pressed by uniaxial pressing with a disk-type 36 mm mold at 25 MPa. The pressed body was heated at a rate of 5°C/min to remove the binder through burning at 600°C; pressed green body was then further heated at a rate of 5°C/min to the sintering temperatures of 1400°C, 1500°C, and 1600°C for sintering for 1 h at each temperature under atmospheric conditions. Subsequently, the pressed green body was cooled at the cooling rate of 5°C/min. To investigate the density depending on the MnO2 content, the bulk density and the open porosity of the specimens prepared at different mixing ratios were measured by Archimedes principle. The total porosity (Φtotal) was calculated by using the bulk density (ρ) as in Equation (1):
The theoretical density (ρo) was calculated from the composition of the raw material powder by using the rule of mixture. The close porosity (Φclosed), the difference of the total porosity (Φtotal), and the open porosity (Φopen) were calculated using Equation (2):
In addition, to verify the change of the shrinkage rate after sintering, the linear shrinkage rate was measured using Vernier callipers. The pore size and the pore distribution of the porous alumina were measured by a mercury porosimeter (AutoPore IV 9510, Micromeritics, USA). The permeability of a porous alumina specimen (diameter: 1.46 cm, thickness: 0.35 cm) was measured by using capillary flow porosimetry (CFP-1200-AEL, Porous Materials Inc., USA). The shape of the powder and the pore structure of the specimens were observed using a scanning electron microscope (SEM, JSM-6610, Jeol, Japan). The flexural strength of the porous alumina was measured by processing the specimens in dimensions of 3 mm × 4 mm × 30 mm. The three-point flexural strength was measured with a measurement length of 20 mm by using a flexural strength tester (RB302, Unitech, R&B, Korea). The electric resistance was measured according to the volume resistance of the specimen after sintering. To measure the electric resistance, the specimens were processed in dimensions of 20 mm × 20 mm × 2 mm using a high-resistance meter (4339B, Agilent Technologies, USA) and applying a (+) bias of 100 V at 25°C.
Additionally, to verify the final phase, a comparative sample for XRD measurement was prepared. To increase the reactivity, the content of the added materials was increased. The raw material used for the preparation of the comparative sample was alumina powder of Al2O3 #600 Mesh (~ 8.93 μm, 99.51% white aluminium oxide grit, Kramer Industries, USA). The preparation was performed under the same conditions described above. TiO2 (99.5% Sigma-Aldrich, USA) was added at 16.65 wt%; MnO2 (99% Sigma-Aldrich, USA) was added at 33.35 wt%. With regard to the composition of the comparative sample, the ratio of MnO2 to TiO2 was 2 : 1, which was the same as the ratio of MnO2 to TiO2 in M4. To verify the generation of a new phase by the addition of the metal oxides to the alumina, and to verify the final phase, an X-ray diffractometer (D/Max 2500 V/PC, Rigaku Corporation, Japan) was used at a scan range of 20° ~ 80° (2θ) and a scan rate of 0.02° [2θ/sec.].
3. Results and Discussion
3.1. Mechanical properties of porous alumina
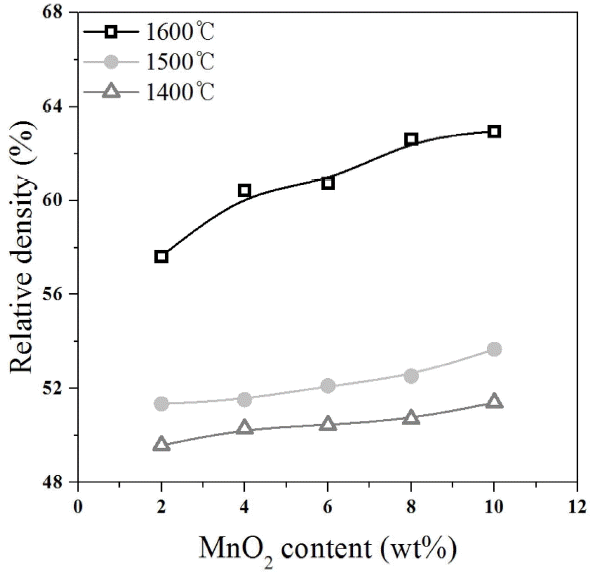
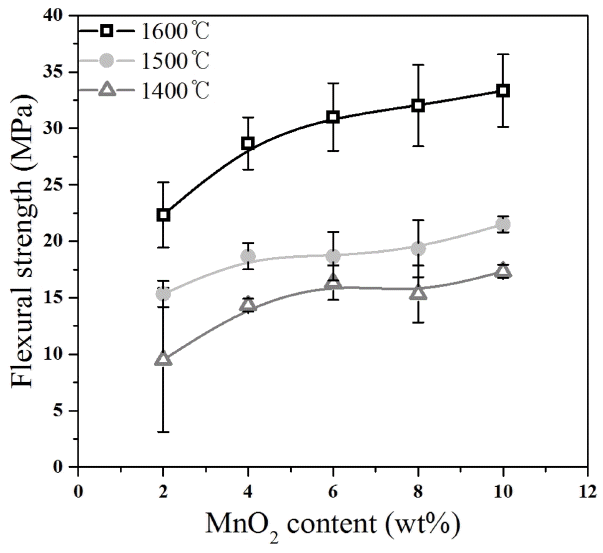
Table 2 shows the compositions used in the experiment. At a constant TiO2 content of 2 wt%, the content of MnO2 was varied to investigate the effect of the MnO2 content on the relative density and strength. According to the MnO2 content, the specimen with MnO2 content of 2 wt% was denoted as M2; specimen with 4 wt% was denoted as M4; specimen with 6 wt% was denoted as M6; specimen with 8 wt% was denoted as M8; and specimen with 10 wt% was denoted as M10. The green bodies prepared at individual compositions were sintered at 1400°C, 1500°C, and 1600°C for one hour at each sintering temperature, and then the relative density, linear shrinkage rate, and flexural strength of the specimens were measured. Fig. 1 shows the relative density of the specimens depending on the sintering temperature and the MnO2 content. The relative density increased as the sintering temperature increased. In particular, the relative density of the specimens sintered at 1600°C was higher than that of the specimens sintered at 1400°C and 1500°C. Thus, the sintering temperature for the alumina powder was determined to be 1600°C in the present experimental study. The strength of the porous alumina is expected to be higher if material is sintered at 1600°C than at 1400°C or 1500°C. The relative density increased as the MnO2 content increased. The M10 specimen with the highest MnO2 content of 10 wt% sintered at 1600°C showed the highest relative density of about 63%, while the M2 specimen with the lowest MnO2 content of 2 wt% sintered at 1600°C showed a relative density of about 57%. Sintering is a process in which necks are formed, crystal grains grow, and volume is shrunk by diffusion through the grain boundary and through the crystal grain lattices at the sintering temperature.8) Therefore, the degree of sintering may be estimated on the basis of the linear shrinkage rate. Fig. 2 shows the measured linear shrinkage rate. The linear shrinkage rate of the specimens sintered at 1400°C and 1500°C was less than 1%, indicating that almost no sintering occurred at these temperatures. On the contrary, the linear shrinkage rate of the specimens sintered at 1600°C was about 3% to 7%, indicating that sintering occurred more actively at 1600°C than at 1400°C and 1500°C. Generally, the linear shrinkage rate of a dense ceramic materials after sintering is about 10% to 20%.9) Therefore, porous alumina, showing a lower linear shrinkage rate than that of dense ceramic materials, may have a lower sinterability. With regard to the effect of MnO2 content on the sintering of the alumina, the linear shrinkage rate increased as the MnO2 content increased. The linear shrinkage rates of the specimens sintered at 1600°C were about 2.70% in M2 and about 7.38% in M10. According to the references, MnO2 added to alumina enhances the sintering of alumina,10-12) and the liquid phase formed by the addition of the MnO2 helps in alumina particle transportation during sintering.13-15) Therefore, the MnO2 added to alumina might have helped the sintering of the alumina. Fig. 3 shows the flexural strength depending on the sintering temperature and the MnO2 content. As the sintering temperature increased, the strength of the specimens sintered at 1600°C was higher than that of the specimens sintered at 1400°C and 1500°C. In addition, as the MnO2 content increased, the flexural strength increased. In particular, the flexural strength of the specimens sintered at 1600°C greatly increased as the MnO2 content increased. The flexural strength of the M10 specimen with the highest MnO2 content was about 36 MPa, and that of the M2 specimen with the lowest MnO2 content was about 25 MPa. The trend in the flexural strength was similar to that of the relative density: The flexural strength increased as the sintering temperature and the MnO2 content increased. Since the added MnO2 may have induced densification of the alumina and helped the neck formation to form a solid structure, the addition of MnO2 to alumina may increase the mechanical strength by increasing the relative density during sintering.
3.2. Pore structure of porous alumina
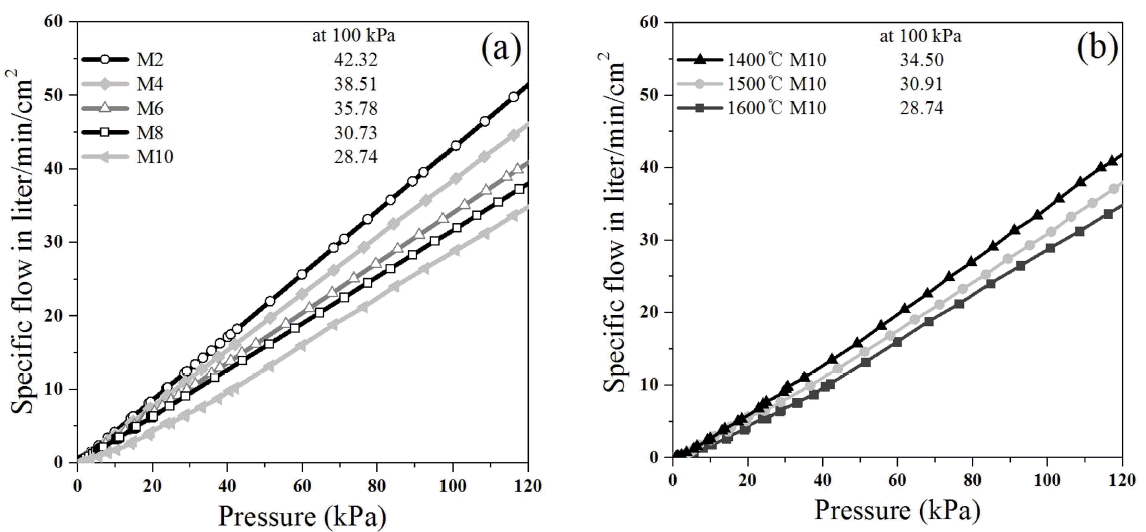
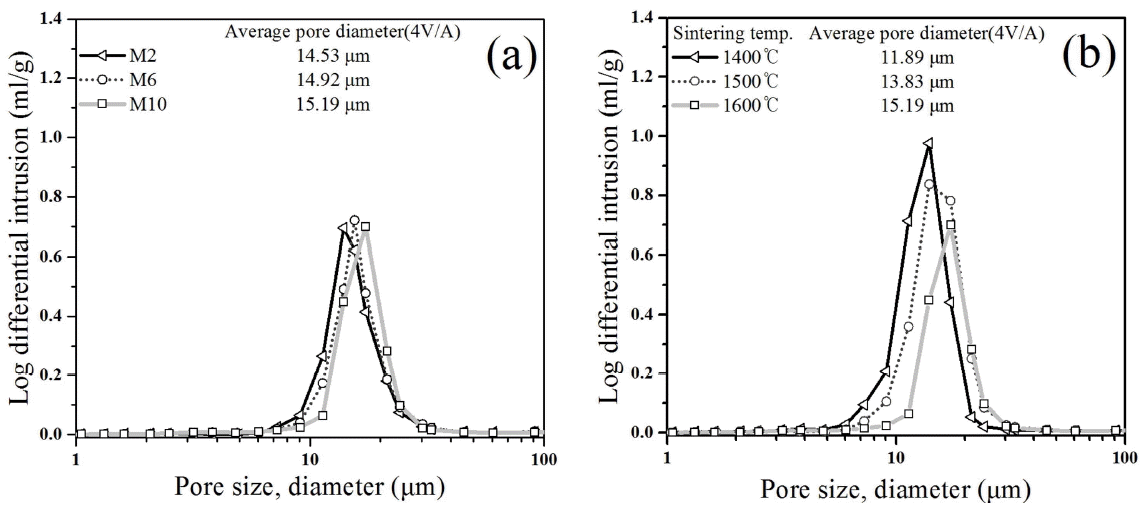
A vacuum chuck requires a high permeability to strongly adhere to a substrate through vacuum suction. In the present study, a coarse alumina powder was used to prepare porous material having a high permeability. As shown in Table 1, the mean particle diameter d(50) was 33.43 μm. Fig. 4 provides a microscopic image of the commercial alumina powder used in the present experiment, indicating that the particles were irregular polyhedrons with angular shapes. The angular shape of the powder may give a low filling ratio but may be helpful in increasing the permeability after sintering. Fig. 5 provides SEM images of the microstructure of the porous alumina-based ceramics. The fracture surface of the sintered body was observed after one hour of sintering at 1600°C. Fig. 5(a) shows the fracture surface of the M2 specimen; 5(b) shows specimen M6; and 5(c) shows specimen M10. According to increase of the MnO2 content, microstructure is showing an increase in the liquid phase, but it does not show a significant difference. The microstructure of the porous alumina prepared using the coarse alumina powder showed a number of large pores. These large pores may have been produced by the large open pores formed between the large powder particles after sintering and may contribute to the increase of the permeability. The permeability was measured to investigate the effect of the MnO2 content on the permeability, as shown in Fig. 6. Fig. 6(a) shows the permeability of sintered bodies prepared by varying the MnO2 content and sintering at 1600°C for 1 h. As the MnO2 content increased, the permeability decreased: the permeability of M2, with the lowest MnO2 content of 2 wt%, was very high at 42.32 liter/min/cm2 under 100 kPa, corresponding to one atmospheric pressure, while the permeability of M10, with the highest MnO2 content of 10 wt%, was 28.74 liter/min/cm2 under the same pressure. This result suggests that the MnO2 content may have affected the permeability. Table 3 shows the permeability depending on the sintering temperature and the MnO2 content, indicating that the total permeability decreased as the MnO2 content increased. Fig. 6(b) shows the effect of the sintering temperature on the permeability, which was measured with the prepared M10 specimens by varying the sintering temperature. The result shows that the permeability decreased as the sintering temperature increased. As the sintering temperature increased, the relative density decreased, which resulted in a decrease of the total porosity and a decrease of the permeability. However, not only the porosity but also the pore size affects the permeability. To verify the pore size distribution and the mean pore size, the mercury porosimeter was used, as shown in Fig. 7. Fig. 7(a) shows the results of pore size distribution and mean pore size of the sintered body prepared by varying the MnO2 content and sintering at 1600°C for 1 h. The mean pore size, shown in Fig. 7(a), increased as the MnO2 content increased. Generally, the permeability increases as the pore size increases. However, in the present study, the porosity decreased as the MnO2 content increased, indicating that the permeability of the porous alumina was affected more by the porosity than by the pore size. Fig. 7(b) shows the results of pore size distribution and mean pore size for the M10 specimens, with the highest MnO2 content of 10 wt%, prepared by varying the sintering temperature. The mean pore size increased as the sintering temperature increased. The mean pore size may have increased along with the pore size growth during sintering, where in the increase of the sintering temperature, one of the driving forces of sintering, removes small pores but makes large pores larger, reducing the specific surface area. As shown in Fig. 7(a), a comparison of the M6 and M10 specimens of different MnO2 content prepared by sintering at 1600°C indicates that the relative density decreased by about 3% and the permeability also decreased due to the increase of the MnO2 content. On the contrary, as shown in Fig. 7(b), comparison of the M10 specimens sintered at 1500°C and 1600°C shows that the porosity decreased by about 10% as the sintering temperature increased, but the permeability did not significantly decrease, in contrast to the expectation. According to the experimental results, the mean pore size of the 1500°C M10 sintered specimen was 13.83 μm, and that of the 1600°C M10 sintered specimen was 15.19 μm, indicating that the mean pore size increased by about 1.36 μm. This may be because small pores were removed but open pores became larger as the sintering temperature increased. Therefore, despite the decrease of the porosity, the permeability increased, probably due to the increase of the pore size. To analyze the effect of the porosity on the permeability, the variation of the porosity depending on the increase of the sintering temperature and the MnO2 content was measured, with results as shown in Table 3. In the cases of the specimens sintered at 1400°C, as the MnO2 content increased, the total porosity did not significantly change, but the open porosity decreased and the closed porosity increased. On the contrary, in the cases of the specimens sintered at higher temperatures of 1500°C and 1600°C, as the MnO2 content increased, the total porosity and the open porosity decreased and the closed porosity increased. This suggests that the total porosity may decrease with the increase of the sintering temperature but the open porosity and the closed porosity may be dependent on the MnO2 content. In contrast to general solid phase sintering, the MnO2 added to alumina may have formed a liquid phase to enhance the sintering of the alumina but may have closed some small pores during the sintering. Thus, as the MnO2 content increased, the quantity of the liquid phase may also have increased, resulting in an increase of the closed porosity. In addition, the closed pores formed by the liquid phase may have decreased the permeability by preventing the gas flow inside the porous material. The porous alumina prepared in the present experiment showed a permeability of about 28 ~ 42 liter/min/cm2 under 100 kPa, corresponding to about one atmospheric pressure. The permeability of the porous alumina prepared in the present study was higher than that of the commercialized vacuum chucks (about 18 liter/min/cm2 under the same pressure). Therefore, the porous alumina prepared in the present study may be applied to parts requiring a high permeability, such as air bearings and vacuum chucks for semiconductor processes.16)
3.3. Electric resistance of porous alumina depending on MnO2 and TiO2 addition
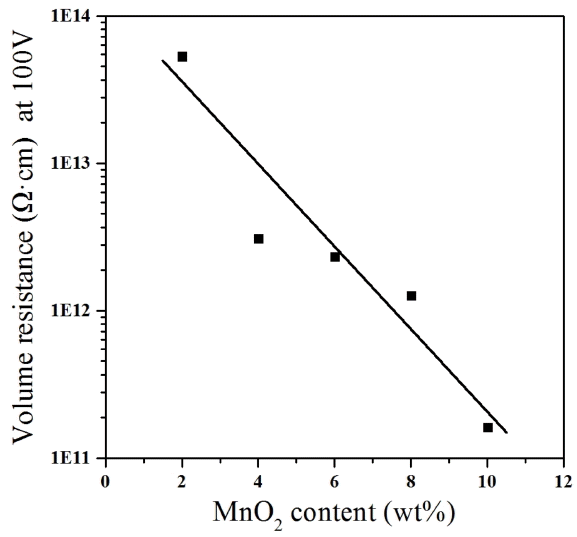
The present experiment was focused on lowering the electric resistance of alumina in order to apply porous alumina to a vacuum chuck, a semiconductor manufacturing device. The generally known volume resistance of high-purity alumina is very high at about 1.7 to 7.2 × 1014 Ω·cm. The electric properties of alumina are affected by the added materials. The volume resistance of alumina with high impurity content is known to be lower than that of high-purity alumina.17) Therefore, in the present experiment, metal oxides MnO2 and TiO2 were added to lower the electric resistance of the porous alumina. First, to verify the variation of the electric resistance depending on the addition of the metal oxides, the volume resistance of alumina was measured using a high-resistance meter; the measurement results are shown in Fig. 8. As the MnO2 content increased, the volume resistance decreased. The highest volume resistance of about 5.3 × 1013 Ω·cm was found in the M2 specimen, having the lowest MnO2 content of 2 wt%, while the lowest volume resistance of about 1.6 × 1011 Ω·cm was found in the M10 specimen, having the highest MnO2 content of 10 wt%. Therefore, the addition of MnO2 and TiO2 to the alumina may effectively decrease the electric resistance. The volume resistance of the alumina samples having the compositions used in the present experiment was lower than that of generally known alumina. Therefore, the alumina in the present study may be used to avoid the problem of device substrate damage caused by static electricity due to the high volume resistance of alumina. As described above, the electric resistance of alumina may be controlled by adding a metal oxide; there may be two possible reasons that the oxide can control the resistance. First, the decrease of the electric resistance of alumina by the addition of a metal oxide may be because of excess electrons generated by a non-stoichiometric equation representing the partial solid solution of the metal oxide in alumina.
As shown in Equation (3), TiO2 may reduce the specific resistance by producing excess electrons as Ti+4 substitutes at the Al+3 site, and thus the volume resistance of alumina may be reduced by the addition of TiO2.18,19) It has been reported that TiO2 is soluble to alumina at a rate of 0.27 wt% in the temperature range of 1300 to 1700°C.20) Since the sintering temperature range of the present experiment was 1400 to 1600°C, the added TiO2 may have undergone partial solid solution. Therefore, the electric resistance of the alumina may have been decreased by the excess electrons produced by the addition of TiO2.
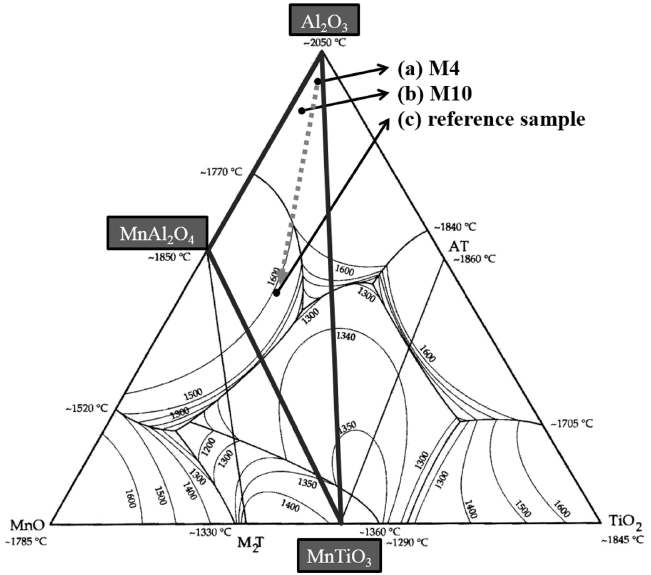
Second, the decrease of the electric resistance of alumina by the addition of a metal oxide, such as MnO2 and TiO2, may be because of the formation of a secondary phase having a lower electric resistance, which results in a decrease of the electric resistance of the alumina. Fig. 9 is the phase diagram of the Al2O3-TiO2-MnO system as reported by M. C. Moreira and A. M. Segadães,21) indicating that compositions of the present experiment are the compositions at which Al2O3, MnTiO3, and MnAl2O4 may coexist in a thermodynamic equilibrium. Fig. 10 shows the final phase of the M4 (a) and M10 (b) compositions, identified by XRD analysis. The final phase of M4 was found to be Al2O3 and MnTiO3, but no peak corresponding to MnAl2O4 was found. In the M10 composition having a MnO2 content of 10 wt%, Al2O3 and MnTiO3 were identified. A peak for MnAl2O4 was expected to be found in the XRD result of the M10 composition, which has a higher MnO2 content than that of the M4 composition. However, the peak intensity of MnAl2O4 was too low to accurately identify. Therefore, for clearer identification, a comparative sample was prepared. The comparative sample was prepared at the same MnO2-to-TiO2 ratio of 2 : 1 as used in the M4 sample, but the alumina content was decreased to 50 wt%. The same MnO2-to-TiO2 ratio of 2 : 1 as in the M4 composition was chosen because no peak for MnAl2O4 was found in the XRD result, although the presence of MnAl2O4 was expected. The alumina content was decreased to 50 wt% to increase the reactivity by increasing the content of the added materials. The comparative sample was prepared using an alumina powder of small particle size. The mean particle diameter of the alumina powder (d(50)) was 8.93 μm. An alumina powder of such a small particle size was used to increase the number of contact points between the alumina and the added materials due to the large specific area of the small particles, because a chemical reaction basically occurs on a contact surface. Therefore, the use of alumina powder of such a small particle size was expected to result in a large peak for the secondary phase. The compositions of the comparative sample correspond to the composition at (c) of the phase diagram shown in Fig. 9. Fig. 10(c) shows the XRD result of the comparative sample, in which peaks for Al2O3, MnTiO3, and MnAl2O4 can be found. In particular, the peak for MnAl2O4, which was difficult to clearly identify in the M4 and M10 compositions, was identified. Therefore, it is assumed that MnAl2O4 may have affected the electric resistance of the alumina. When a coarse alumina powder is used, as in the present experiment, the specific surface area is smaller than that of the alumina powder, which has a smaller particle size, resulting in the production of a smaller amount of MnAl2O4. Therefore, the intensity of the MnAl2O4 peak was weaker in the XRD results of the M4 and M10 compositions. According to previous reports, the electric resistance of MnAl2O4 is about 1.0 × 103 Ω·cm, which is lower than that of alumina;22) the electric resistance of MnTiO3, produced by the sintering of MnO2 and TiO2 together, is about 1.8 × 107 Ω·cm.23) Hence, sintering of the alumina containing added MnO2 and TiO2 may have resulted in the formation of MnTiO3 and MnAl2O4, with the secondary phases having a significantly lower electric resistance than that of the alumina, and thus the electric resistance of alumina was lowered.
The electric resistance may have also been decreased by the solid-solution of TiO2, but specific evidence of the solid-solution of TiO2 could not be found in the experimental results. However, the possible presence of a secondary phase having a low electric resistance was verified in the phase diagram of the Al2O3-TiO2-MnO system reported by M. C. Moreira and A. M. Segadães,21) and an XRD peak corresponding to a secondary phase was also clearly found. Therefore, the decrease of the electric resistance by the formation of a secondary phase having low electric resistance may be considered an important mechanism.
4. Conclusions
In the present study, an experiment was performed to prepare a semi-conducting porous alumina ceramic that may be applied to a vacuum chuck for semiconductor processes. Porous specimens were prepared by varying the MnO2 content of the alumina containing 2 wt% of TiO2, and the following conclusions were obtained by measuring the mechanical properties, pore structure, and electric resistance of the porous specimens. The coarse alumina powder formed large open pores, which resulted in an increase of the permeability. The low mechanical strength of the porous material was improved by increasing the density of the porous alumina during sintering by adding MnO2 to the alumina. In addition, metal oxides were added to the alumina to form a secondary phase having a low electric resistance, such as MnTiO3 and MnAl2O4, verifying the possibility of preparing a porous alumina material having a low volume resistance. In the porous alumina, the relative density increased as the MnO2 content increased. The increase of the relative density also increased the flexural strength but decreased the permeability and the electric resistance. The flexural strength of the M10 specimen with the highest MnO2 content of 10 wt% was about 36 MPa and showed the lowest volume resistance of 1.63 × 1011 Ω·cm. The mean pore size of the porous alumina was about 15 μm. The permeability was high at about 28 liter/min/cm2 under the gas flow pressure difference of 100 kPa.