Modeling, Preparation, and Elemental Doping of Li7La3Zr2O12 Garnet-Type Solid Electrolytes: A Review
Article information
Abstract
Recently, all-solid-state batteries (ASSBs) have attracted increasing interest owing to their higher energy density and safety. As the core material of ASSBs, the characteristics of the solid electrolyte largely determine the performance of the battery. Thus far, a variety of inorganic solid electrolytes have been studied, including the NASICON-type, LISICON-type, perovskite-type, garnet-type, glassy solid electrolyte, and so on. The garnet Li7La3Zr2O12 (LLZO) solid electrolyte is one of the most promising candidates because of its excellent comprehensively electrochemical performance. Both, experiments and theoretical calculations, show that cubic LLZO has high room-temperature ionic conductivity and good chemical stability while contacting with the lithium anode and most of the cathode materials. In this paper, the crystal structure, Li-ion transport mechanism, preparation method, and element doping of LLZO are introduced in detail based on the research progress in recent years. Then, the development prospects and challenges of LLZO as applied to ASSBs are discussed.
1. Introduction
To meet the increasing energy demand, more efficient and safer electrochemical energy storage has become a hot spot in related fields. With their high energy density and favorable cyclic stability, Li-ion rechargeable batteries (LIBs) stand out from other electrochemical energy storage devices and have been widely used in power supplies such as electrified transportation and portable electrical devices,1,2) However, despite their high lithium-ion conductivity, conventional lithium-ion batteries with liquid electrolytes may result in excessive charging, internal short circuiting, and other anomalies that lead to serious consequences such as leakage, self-ignition or even explosion.
In comparison to LIBs, ASSBs with solid electrolytes show great prospects and can be considered as the next generation of devices for energy storage on account of their comparatively excellent properties: higher safety, longer cycle life, higher energy density and power density, fewer requirements for packaging, lower self-discharge rate, state-of-charge monitoring circuits, and no memory effect.3–5) To meet the requirements for the successful fabrication and commercialization of all-solid-state lithium secondary batteries, solid electrolyte which is the key influencing component, should have a wide electrochemical window, high lithium-ion conductivity, and good chemical and electrochemical stability for lithium.6)
Nowadays, the mainly researched solid electrolytes are polymer electrolytes and inorganic ones. As the most representative polymer electrolytes, poly(ethylene oxide) (PEO)- based electrolytes show very good solubility for lithium salts as well as good flexibility and tensile shear properties. However, their low ionic conductivity at ambient temperature and narrow electrochemical window are insufficient for meeting practical requirements.7) By contrast, inorganic electrolytes have many advantages including relatively high ionic conductivity, high-level safety, and wide electrochemical window. The research studies of inorganic electrolytes have mainly concentrated on NASICON, garnet, LISICON, perovskite, LiPON, and sulfide electrolytes, but most of them cannot meet all of the needs above. NASICON- type electrolytes, which are stable in air and water and have high lithium-ion conductivity, allow the migration of the charge carriers in all three dimensions. A typical NASICON-type electrolyte Li1.3Al0.3Ti1.7(PO4)3 whose skeleton is most suitable for ionic migration possesses a high ionic conductivity of 7 × 10−4 S/cm at 25°C.8) However, these kinds of electrolytes still show drawbacks of bad interfacial contact as well as being unstable with Li metal owing to facile Ti4+ reduction.9,10) With the same drawbacks as the NASICON-type,11) the perovskite-type electrolyte can achieve a grain conductivity close to 10−3 S/cm, but the total conductivity is relatively low owing to the high boundary resistance.12,13)
Existing scientific research shows that the ionic conductivity has a tangible correlation with the formation enthalpy.14) LISICON-type electrolytes can show great stability even with long cycling and high rate capability,15) but in contrast to the above two types of electrolytes, the LISICON-type ones have relatively low lithium-ion conductivity that decreases further with time. These electrolytes are also unstable to Li metal and atmospheric CO2.9,16) Although LiPON-type electrolytes show great compatibility with positive and negative poles, high mechanical stability, and a broad electrochemical stability window, the low conductivity of about 2.3 × 10−6 S/cm at room temperature cannot be ignored in applications. 17,18)
The sulfide-type electrolyte Li9.54Sil.74P1.44S11.7Cl0.3 has a lithium-ion conductivity of about 10−2 S/cm at ambient temperature. 19) These electrolytes also show good performance in thermostability and softness. Nonetheless, the bad contact and stability with both poles as well as the release of H2S or other harmful gas after reacting with water and oxygen in the air cause adverse impacts on the further research and applications of sulfide-type electrolytes.9,20)
The typical garnet-type material LixLa3M2O12 (M = Ta, Nb, Zr) shows numerous advantages such as high lithiumion conductivity, negligible electronic transport, good thermal and chemical stability against Li metal, and a wide electrochemical window. With better performance, the traditional garnet-type electrolyte Li7La3Zr2O12 (LLZO) shows high Li-ion conductivity of 0.1–1 mS/cm at ambient temperature and a wide electrochemical window of above 5 V after adulteration.21) Since 2007, garnet-type Li7La3Zr2O12 was firstly reported by Murugan using a conventional solid-state reaction with an ionic conductivity of 3.0 × 10−4 S/cm,22) many studies of LLZO have been done on account of its high conductivity and the higher possibility of synthesizing the ideal electrolyte. In addition, many studies were conducted to obtain electrolytes with excellent properties by modifying LLZO.23–27) As a result of a comprehensive comparison, LLZO is considered one of the most promising electrolytes to meet the requirements of ideal electrochemical energy storage devices.28)
In this review, we first analyze the crystal structure, phase transition process, and lithium ion migration mechanism in LLZO. Then, different synthesis methods and their corresponding characteristics are introduced in detail. Finally, we summarize some research progress in the element doping of LLZO in recent years and present some perspectives.
2. Crystal Structure and Lithium Diffusion Mechanism of Li-stuffed Garnet
Generally, lithium garnet shows two crystal structures: tetragonal and cubic phase. The two garnet phases have the same framework structure but differ in lithium distribution, which strongly affects the lithium diffusion mechanism and electrochemical performance of the two garnet phases. For a better understanding of lithium garnet, this section introduces the crystal structure of the two garnet phases, after which the theoretical and experimental approaches to the phase transition process and lithium migration mechanism are discussed.
2.1. Crystal structure
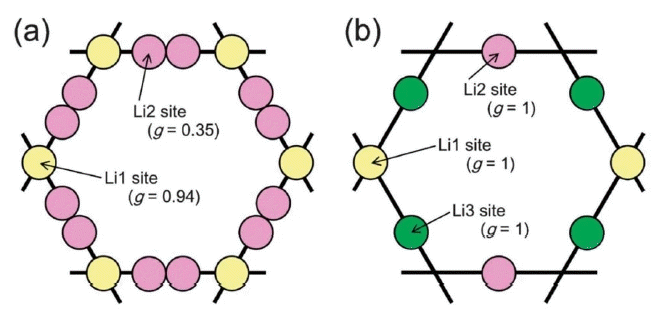
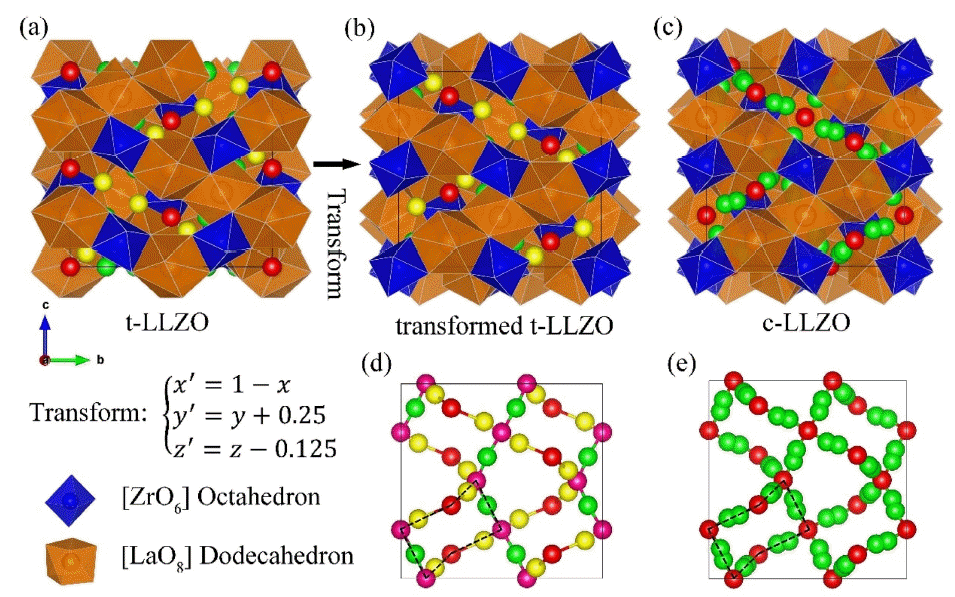
A fast Li+ conductor with a garnet-like structure was first reported by Prof. Werner Weppner’s Group,29,30) The lithium garnet family exhibits the general formula of A3B2C3O12 (A = La3+, Y3+, Nd3+; B = Te6+, Ta5+, Nb5+, Zr4+), where A and B are eight- and six-coordinated sites. The framework structure is composed of edge-sharing [AO8] dodecahedron and [BO6] octahedrons. Lithium ions occupy the interval tetragonal and octahedral sites. These lithium polyhedrons interconnect to form a lithium loop structure as shown in Fig. 2, which is the basic lithium diffusion pathway in a lithium garnet solid electrolyte.31) Commonly studied lithium garnets contain three to seven per formula and are usually crystallized in a cubic structure with an Ia3̄d space group.30–32) However, Awaka et al.33) found that Li7La3Zr2O12(LLZO) prefers crystallization in a tetragonal phase with I41/acd at room temperature and exhibits a lithium ionic conductivity that is two orders of magnitude lower than that of the cubic phase. A less conductive tetragonal phase also exists in other lithium garnets with seven Li+ per formula, such as Li7La3Sn2O12 34) and Li7La3Hf2O12.35) The crystal structures of tetragonal LLZO (t-LLZO) and cubic LLZO (c-LLZO) are shown in Fig. 1. t- and c-LLZO have the same framework structure but differ in their lithium distributions. In the cubic phase, lithium disordering is observed in partially occupied c-24d (tetragonal site) and c-96h (octahedral site) sites. In the tetragonal phase, t-8a (tetragonal site), t-16f, and t-32g (octahedral site) are fully occupied by lithium ions, leaving t-16e (tetragonal site) unoccupied. This results in the complete ordering of lithium ions and a Li-Li distance longer than 2.5 Å.31)
2.2. Phase transition
Depending on the synthesis and simulation methods, the phase transition temperature varies from 450 to 1000 K.36–40) Bernstein et al.37) stated that the tetragonal phase stability gains from the lithium-ion sublattice ordering and volume-preserving tetragonal distortion, which increase the Li-Li distance and relieve the internal structural strain. Therefore, the introduction of a lithium vacancy is necessary for the stabilization of the cubic phase. Adams et al.40) performed a molecular dynamic simulation and in-situ XRD to study the phase transition in lithium garnets. The results, as shown in Fig. 3, confirmed that the cubic phase can be stabilized by pentavalent doping, which increases the lithium vacancy concentration and reduces the local lithium-ion ordering.
From ordering to disordering, the phase transition from t- LLZO to c-LLZO is accompanied with a lithium redistribution among all lithium sites. Bernstein et al.37) suggested that lithium redistribution is simply a shift from t-8a to t- 16e based on the unchanged occupancy of c-24d (c-24d = t- 8a + t-16e). Klenk et al.38) studied the evolution of the cage occupancy for four different lithium sites (t-8a, t-16e, t-16f, and t-32g) with temperature. The results are shown in Fig. 4. They concluded that lithium redistributes among all tetragonal sites. In addition, the phase transition initiates at the t-8a site with the cooperation of the t-32g site. Chen et al.41) calculated the defect energy of four lithium vacancy models and concluded that the lithium redistribution is determined by the site energy preference.
The lithium vacancy located at the t-32g site exhibits the lowest energy among the t-8a, t-16f, and t-32g sites, which leads to the initiation of Li redistribution on the t-32g site. Then, lithium ions at the t-8a and t-16f sites participate in the redistribution one after another. In conclusion, the phase transition from t-LLZO to c-LLZO is a site-energy-preference- determined, entropy-driven process, the physical expression of which is the redistribution of lithium ions from the t-32g, t-8a, and t-16f to t-16e sites.
2.3. Lithium diffusion
The difference in lithium distribution between t-LLZO and c-LLZO results in reduced lithium diffusion properties of t-LLZO compared to c-LLZO. Many research studies have been carried out to reveal the relationship between the lithium arrangement and the lithium diffusion mechanism. Meier et al.42) performed first-principles-based calculations to study the lithium diffusion mechanism in both t-LLZO and c-LLZO as shown in Fig. 5. The results suggested that lithium diffusion exhibits a fully collective motion in t-LLZO and single-ion jumps in c-LLZO. The latter mechanism exhibited a relatively lower energy barrier. Jalem et al.43) studied the lithium diffusion in c-LLZO by molecular dynamic calculation. A concerted migration mechanism for lithium diffusion was also found in c-LLZO.
A classical molecular dynamic simulation performed by Klenk et al.38,39) suggested that lithium diffusion exhibits oscillations in low-temperature t-LLZO and “structured diffusion” in high-temperature c-LLZO. The researchers also calculated the Haven ratio (the ratio of self-diffusion vs. collective diffusion), which increased from 0.05 to 0.35 when the temperature increased from 700 to 1400 K. The results of the Haven ratio calculation illustrated that the collective diffusion exists in both t- and c-LLZO, but varies in degree. By combining a lithium local structure and the evolution of the site occupation with temperature, Chen et al.41) found a temperature-dependent lithium migration pathway. Lithium ions exhibited uniformly three-dimensional diffusion in c-LLZO, which is the same as in other reports. By contrast, in t-LLZO, an unusual two-dimensional-like lithium diffusion mechanism was observed. The decrease of the migration pathway with regard to its dimension is responsible for the low lithium diffusion properties in t-LLZO. Chi Chen et al.44) combined machine learning and information theory to analyze molecular dynamic trajectories, and found that c- LLZO has a higher hopping frequency than t-LLZO. In addition, lithium exhibits uncorrelated Poisson-like diffusion in c-LLZO and correlated diffusion in t-LLZO as shown in Fig. 6. This work introduced a unique perspective in analyzing molecular dynamic results.
3. Synthesis Methods
LLZO can be obtained by different synthesis methods: conventional solid-state reaction, sol-gel method, field-assisted sintering,45–48) coprecipitation,48,49) electrospinning,50) and spray pyrolysis synthesis.51)
3.1. Conventional solid-state reaction
As the most widely used method to prepare LLZO, the conventional solid-state reaction has several advantages: a simple preparation process, suitability for a large yield, low cost, etc. The grinding and sintering process in this method needs to be repeated in order to enhance the performance of products. The purpose of the first round of grinding and sintering is to mix the raw materials and obtain the early LLZO phase, respectively. The purpose of the second round of grinding and sintering is to obtain a uniform particle size and the final product, respectively.
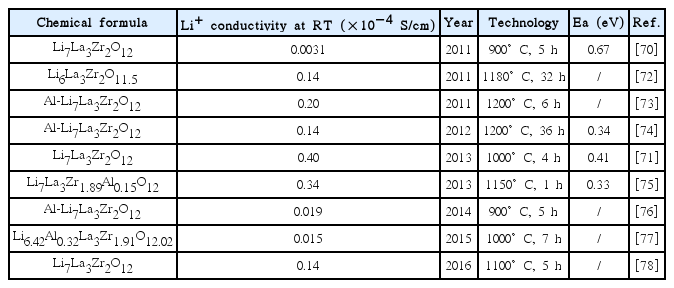
The first report on LLZO was published by Murugan et al. in 2007.30) The author used a conventional solid-state reaction to prepare Li7La3Zr2O12 at 1230°C for 36 h. The Li+ conductivity was 3 × 10−4 S/cm. Many research studies on LLZO were conducted afterward. However, impure phases are introduced to systems easily via the conventional solid-state reaction because the high temperature and long sintering time cause too much lithium loss, and other elements diffuse from the crucible. Kai Liu et al.53) prepared Li6.5La3- Ta0.5Zr1.5O12 in an alumina crucible and LiAlO2 appeared as a second phase since the Al3+ diffused from the alumina crucible to the LLZO. Rangasamy et al.52) prepared Al-doped LLZO with various Li+ concentrations by adding different amounts of lithium sources. However, there was La2Zr2O7 and LaAlO3 in the samples. The XRD of the samples is shown in Fig. 7. To prepare pure LLZO samples, mother powders were placed on an alumina crucible to prevent the pellets from being contaminated by Al3+. Different amounts of lithium sources were adopted to compensate for the lithium loss during calcination.
In addition, in order to obtain LLZO with higher Li+ conductivity and a more stable structure, researchers adjusted the types of raw materials, lithium content, process parameters (sintering temperature, pressure, time, etc.), and various dopants. Allen et al.54) used Li2CO3, La(OH)3, and ZrO2 as raw materials, which were calcined at 1000°C for 3 h to obtain LLZO with a Li+ conductivity of 3.7 × 10−4 S/cm. Its density was as high as 98%, while the activation energy was only 0.30 eV. Lee et al.55) used LiOH, La2O3, and ZrO2 as raw materials. After high-energy ball milling for 12 h, the pellets were calcined at 1125°C for 20 h to obtain LLZO with a Li+ conductivity of 4.9 × 10−4 S/cm at room temperature. Yutao Li et al.56) used 0.6 mol of Ta-doped Zr sites and prepared Li6.4La3Zr1.4Ta0.6O12 in an alumina crucible. The ionic conductivity was 1.0 × 10−3 S/cm. Jianli Gai et al.57) reported on Li7La3ZrNb0.5Y0.5O12 prepared by co-doping Nb and Y on a Zr site, for which the ionic conductivity was 8.29 × 10−4 S/ cm. Xin Guo et al.58) used a Ga-doped Li site to obtain Li6.24Ga0.25La3Zr2O12. The sample exhibited excellent Li+ conductivity as high as 1.46 × 10−4 S/cm at room temperature. Yiqiu Li59) prepared Li6.75La3Zr1.75Ta0.25O12 in different atmospheres and found that O2 contributes to the growth of grains and enhances the Li+ conductivity. A sample sintered in O2 exhibited a maximum Li+ conductivity of 7.4 × 10−4 S/ cm at room temperature.
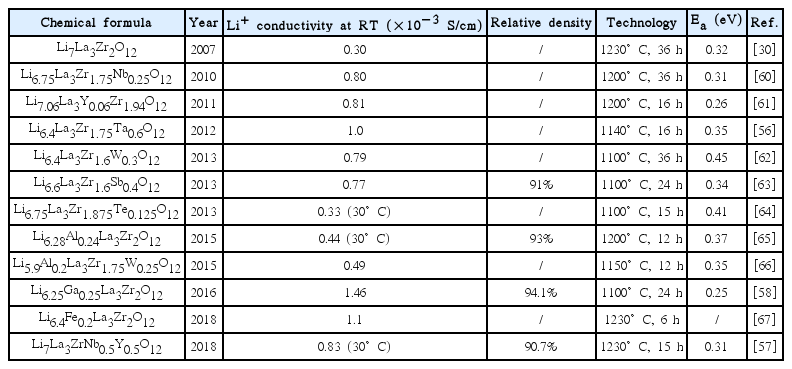
Table 1 summarizes the cubic phase LLZO prepared by the conventional solid-state reaction that was reported.
3.2. Sol-gel method
In order to get LLZO with a smaller particle size, more uniform distribution, and lower sintering temperature, the sol-gel method is usually employed. The sol-gel method can make the components uniformly mix in the liquid phase and react with each other to obtain a uniform precursor powder. Then, the precursor powder will be sintered at a relatively low temperature to prepare a nanosized LLZO powder.68) This is beneficial for the preparation and performance optimization of inorganic solid electrolytes. Xie et al.69) and Kokal70) successfully synthesized pure-cubic-phase LLZO nanopowders by the sol-gel method at a low temperature of 750°C, but the Li+ conductivity was only 10−6 S/cm at room temperature. Sakamoto et al.71) used LiNO3, La(NO3)3·6H2O, and Zr(OH7C3)4 as raw materials and acetic acid as chelating agent. The wet gel was dried by a supercritical drying technique and then sintered at 1000°C for 4 h. The nano-cubic phase LLZO with a grain size of only 260 nm had a high Li+ conductivity (4 × 10−4 S·cm−1) at room temperature. I. Kokal70) prepared LLZO, and the total Li+ conductivity was 3.12 × 10−7 S/cm. Fig. 8 shows SEM micrographs of the LLZO powder precursors calcined at different temperatures. It can be seen that the size of the precursors is on the nanoscale. Reports on the preparation of LLZO by the solgel method in recent years are listed in Table 2.
3.3. Field-assisted sintering method
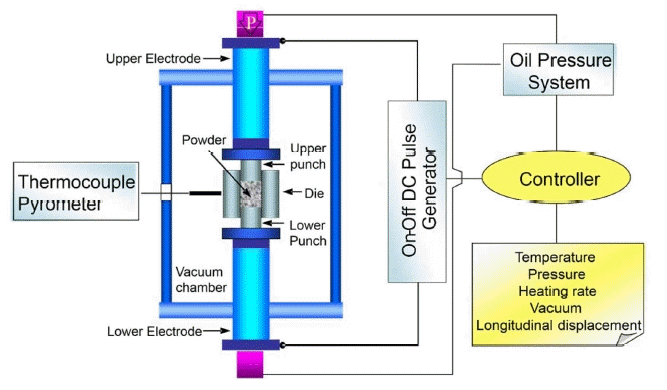
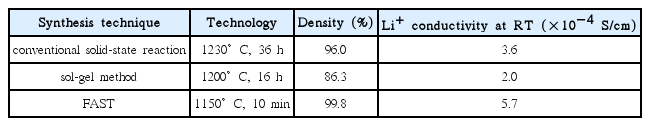
Field-assisted sintering technology (FAST) is a new type of technology for the rapid sintering preparation of LLZO. This process uses uniaxial pressure and passes direct current through a mold. This method is similar to hot pressing (HP). In comparison with hot pressing, where the heat generation occurs externally, the heat is produced internally for FAST. The raw material powder is placed in a mold made of graphite or other materials, and the heat is transmitted by conduction to the powder. Because of the good electrical conductivity of materials used for the mold, high current can be achieved at low voltage. A spark discharge removes impurities from the surface of the particles and activates the surface to promote sintering, which is beneficial to the low temperature, rapid sintering, and densification of the raw material powder.79) Zhang et al.45) used field-assisted sintering technology to prepare LLZO with a high density of 99.8% for the first time at 1100°C. The sintering time was only 10 min, and the Li+ conductivity was 5.7 × 10−4 S/cm at room temperature, which is the highest among the same materials. In order to increase the production scale of the laboratory, Botros et al.46) combined spray pyrolysis (NSP) and field-assisted sintering to reduce the sintering temperature to 950°C. The Li+ conductivity was 3.3 × 10−4 S/cm. Zhang et al.48) prepared the precursor powder of a nano-nuclear- shell structure by the coprecipitation method, and then reduced the sintering temperature to 900°C by field-assisted sintering technology. Finally, they obtained the LLZO with a particle size of 1–3 μm. The Li+ conductivity was 3.32 × 10−4 S/cm at room temperature with a density of 96.5%. Since the sintering time of the field-assisted sintering method is very short, the lithium loss during sintering can be effectively reduced. The density of the material can be improved by applied uniaxial pressure during sintering. Thus, the field-assisted sintering method is particularly suitable for rapidly preparing high-purity, high-density LLZO electrolyte materials. A schematic diagram of FAST is shown in Fig. 9.
SEM micrographs of cross sections of the samples are shown in Fig. 10. These samples were sintered at (a) 1150°C and (b) 1200°C. The SEM images revealed good interfacial bonding between the particles. There were no intergranular fractures and almost no voids.
Table 3 shows a comparison between FAST and the two abovementioned preparation methods. In this table, the advantages of FAST can be clearly seen.
3.4. Other preparation methods
In addition to the abovementioned methods, new preparation methods of LLZO have been studied. These include coprecipitation, electrospinning, and spray pyrolysis. Every method offers its own advantages and is suitable for a specific use. For example, Fig. 11 shows the experimental setup for nebulized spray pyrolysis.80) Nebulized spray pyrolysis (NSP) is a solution-based technique; however, organic solvents can be eliminated as an annealing treatment. The powder size can reach the nanometer scale. Furthermore, the synthesis time is shorter using this NSP technique compared to other techniques. The electrospinning technique was chosen to prepare nanostructured LLZO such as ceramic nanofibers and nanowires. Additionally, the phase transformations can be investigated. However, these methods have not yet matured and cannot meet the large-scale requirements of commercial production. Therefore, researchers are still working on the development of new, efficient, and energy-saving preparation methods that can reduce the cost, sintering temperature, and cycle time while also taking into account the electrochemical performance of LLZO.
4. Element Doping in LLZO
Because of the different diffusion mechanisms, the ionic conductivity of c-LLZO is two orders of magnitude higher than that of t-LLZO. However, the structure of c-LLZO at high temperature is unstable and easily converted into a tetragonal phase.81,82,113) Therefore, in addition to exploring a reasonable preparation method, stabilizing the cubic phase lattice to improve its ionic conductivity at room temperature is another research focus of LLZO.
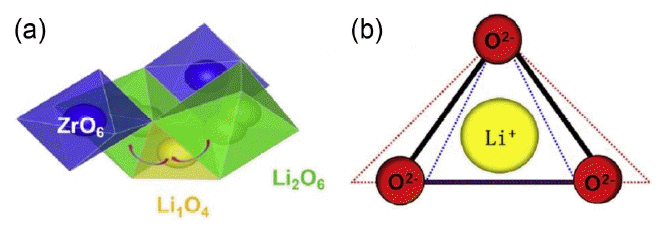
Element doping is one of the most effective ways to stabilize the cubic phase lattice and improve the ionic conductivity. On the one hand, element doping can regulate the concentration of lithium ions to effectively increase the lithium vacancy concentration and the disorder of the lithium ion arrangement in the lattice, thus stabilizing the cubic phase and optimizing the ionic conductivity. On the other hand, the skeleton structure of LLZO lattice can be adjusted by element doping to increase the lithium ion mobility. As shown in Fig. 12, the transport path of Li+ in the LLZO lattice is that Li+ migrates from [Li2O6] octahedron to an adjacent [LiO4] tetrahedron, and then migrates to another adjacent [Li2O6] octahedron. The bottleneck size of the Li+ transport is the size of a triangle composed of three coplanar oxygen atoms of [Li2O6] octahedral and [LiO4] tetrahedral.83)
Taking the Zr site substitution as an example, because [ZrO6] octahedrons and [Li2O6] octahedrons are co-edge-joined, the difference in the ionic radius between the doping element and Zr can change the bottleneck size of the Li+ transport path, which affects the activation energy of lithium ion migration. As a consequence, the bottleneck size of the lithium ion transport has a significant influence on the ionic mobility and ionic conductivity. There are three types of doping and substitution sites for LLZO: Li site, La site, and Zr site. The effect mechanisms of doping at different sites on the ionic conductivity of LLZO are different. The study results of element substitutions for Li, La, and Zr sites in recent years are listed in Table 4.
4.1. Li site substitution
For doping an Li site, researchers usually use Al3+, Ga3+, Fe3+, and other elements to partially substitute the Li element. The substitution of the Li site only affects the lithium ion concentration, and research studies have shown that the concentration and different distributions of lithium ions in LLZO have a crucial influence on its ionic conductivity. Rangasamy et al.84) found that 0.2–0.24-mol Al substitution in LLZO can stabilize the cubic phase, and their ionic conductivities were higher than that of LLZO without Al doping. This phenomenon was quickly confirmed by other researchers who also studied Al-doped LLZO.85,86) Subsequently, Bernstein et al.87) combined density functional theory and molecular dynamics calculations to prove that the introduction of lithium vacancies in a concentration range of 0.4 to 0.5 in the LLZO structural unit is more conducive to the improvement of the ionic conductivity. Jin et al.88) found that Al doping not only changes the Li+ concentration in the sample but also promotes the densification of the sample, resulting in an increase in ionic conductivity. As the Al content increased from 0 to 0.25 wt.%, the density of the sintered sample increased from 2.6 g·cm−3 to 4.5 g·cm−3. Accordingly, the ionic conductivity increased from 1.2 × 10−7 S/cm to 2.0 × 10−4 S/cm.
Zhang et al.47) prepared LLZO ceramic pellets with Li+ concentration of 5.47 mol to 7.37 mol. The relationship between the Li+ concentration and the ionic conductivity was studied by a nuclear magnetic resonance (NMR) test. It was found that when the Li+ concentration exceeded 7 mol, the XRD peak of LLZO broadened, indicating that the cubic phase changed to the tetragonal phase. This is the result of a highly ordered arrangement of lithium ions. When the Li+ concentration was 6.35 mol, the ionic conductivity reached its peak, indicating that a proper reduction in the Li+ concentration (or increase in the lithium vacancy concentration) for LLZO is beneficial to the improvement of ionic conductivity. Therefore, the substitution of nonequivalent cations for Li sites and the use of extra positive charges to promote the formation of lithium vacancies have become research hot spots for improving the ionic conductivity of LLZO.
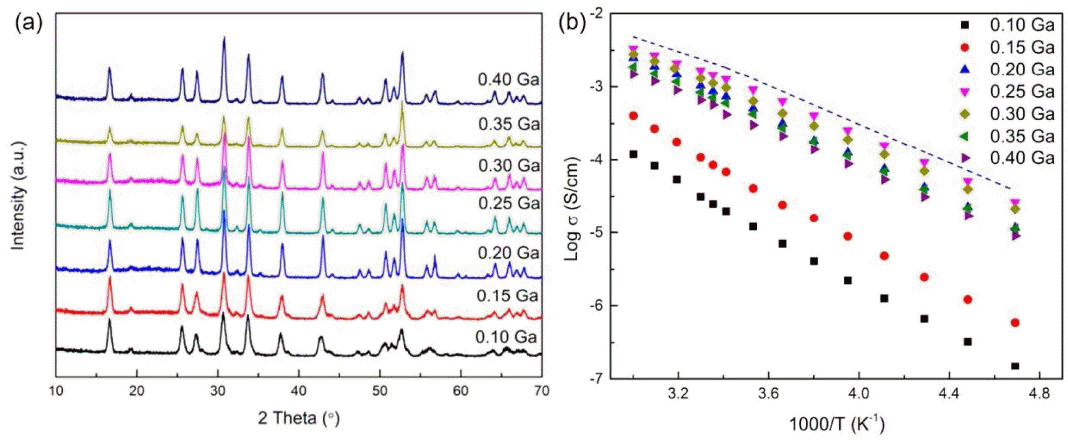
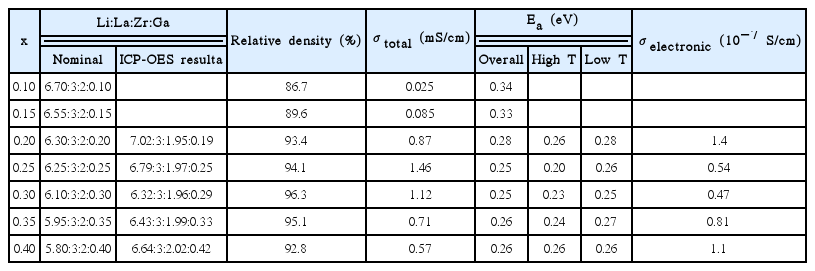
Wolfenstine et al.89) believed that because of the site preference and slightly larger size of Ga than Al, it is highly possible for Ga-doped LLZO to have a lower occupancy fraction on the Li1 (tetrahedral) site than Al-doped LLZO, thus leading to higher ionic mobility and conductivity. Cubic Li6.25La3Zr2Ga0.25O12 powders with a relative density of ~ 91% were synthesized by hot pressing. They had a total Li-ion conductivity of ~3 .5 × 10−4 S/cm, which was slightly higher than that of Al-doped LLZO of a similar composition and relative density. Wu et al.90) prepared a series of Li7-3xGaxLa3Zr2O12 (x = 0.10, 0.15, 0.20, 0.25, 0.30, 0.35, and 0.40) electrolytes via a solid-state reaction. The Li magic angle spinning nuclear magnetic resonance (MAS-NMR) spectra were used to analyze the lithium ion dynamics. Fig. 13(a) shows the X-ray diffraction (XRD) patterns of the Li7-3xGaxLa3Zr2O12 samples. The patterns and lattice parameters match well with the cubic garnet phase. The conductivity of the Li7-3xGaxLa3Zr2O12 samples was measured by the AC impedance technique. and to total conductivity is calculated in Table 5.
Figure 13(b) shows the temperature dependence of the total conductivities of the Li7-3xGaxLa3Zr2O12 samples. It is worth noting that the conductivities of the samples (x ≥ 0.20) with the cubic phase are higher than those of the samples (x < 0.20). A cubic structure is obtained when the Ga concentration exceeds 0.2 per formula unit, while a highest ionic conductivity of 1.46 mS/cm at 25°C was achieved at x = 0.25. A special distribution in which most lithium within LLZO resides at the octahedral 96 h site, away from the central octahedral 48 g site, while the remaining lithium resides at the tetrahedral 24 d site, leads to high lithium-ion mobility as well as high conductivity.
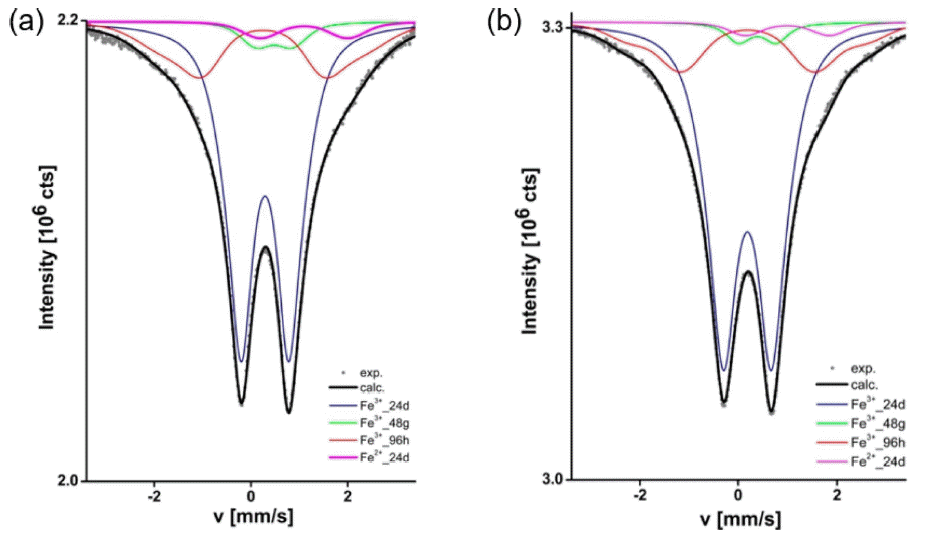
Compared to the doping of Al3+ and Ga3+, doping with other elements such as Fe3+ and Ge4+ has seldom been studied. Rettenwander et al.91–93) found a very high total conductivity of 1.1 mS/cm at room temperature, but the Fe-doped LLZO was not stable vs. lithium metal since it formed a thick tetragonal LLZO interlayer, causing high interfacial impedance. 57Fe Mössbauer spectra of FexLi7–3xLa3Zr2O12 is shown in Fig. 14, which indicates that about 96% of the total iron occurs as Fe3+ and 4% as Fe2+. Hence, Fe3+ seems to act similarly as Al3+ in LLZO which can stabilize the cubic phase of LLZO and promote its ionic conductivity.91)
As shown in Fig. 15 , the Raman spectroscopy of LLZO/Li interface indicates that there are two different phases of LLZO (cubic phase and tetragonal phase) present in the interfacial layer which causes by the contact of cubic LLZO with Li metal. The formation of t-LLZO will lead to an increased interfacial resistance because of the lower ionic conductivity of t-LLZO. Huang et al.94) studied the doping effect of In, Si, and Ge. The researchers found that Si and In doping deteriorates the electrical properties of LLZO, while Ge-doped LLZO presents an enhanced ionic conductivity of 7.63 × 10−4 S/cm at 298 K.
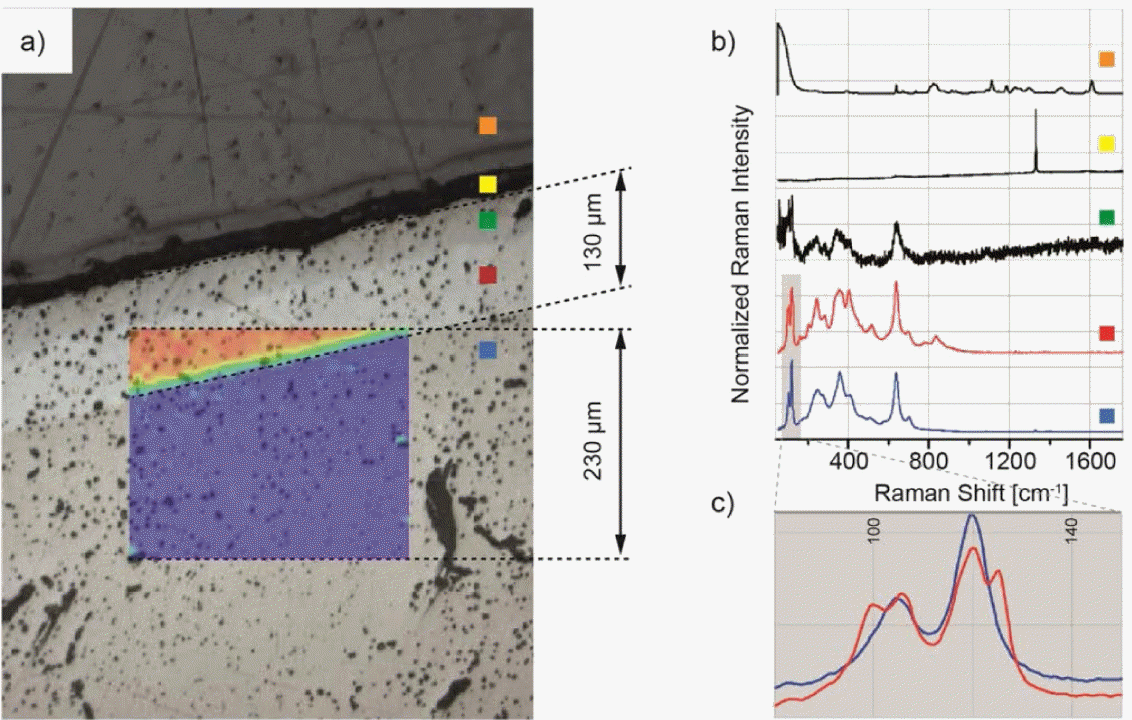
(a) Raman mapping of the Li6.4Fe0.2La3Zr2O12; the picture shows a cross section after the solid electrolyte has been in contact with metallic lithium during cyclic voltammetry. (b) Representative spectra of the different areas of the cross section. (c) Magnification of the shaded area in (b) showing cubic and tetragonal LLZO. Color code: Cubic LLZO (blue), tetragonal LLZO (red), (distorted spectrum of) tetragonal LLZO (green), diamond (yellow), epoxy resin (orange).93)
4.2. La site substitution
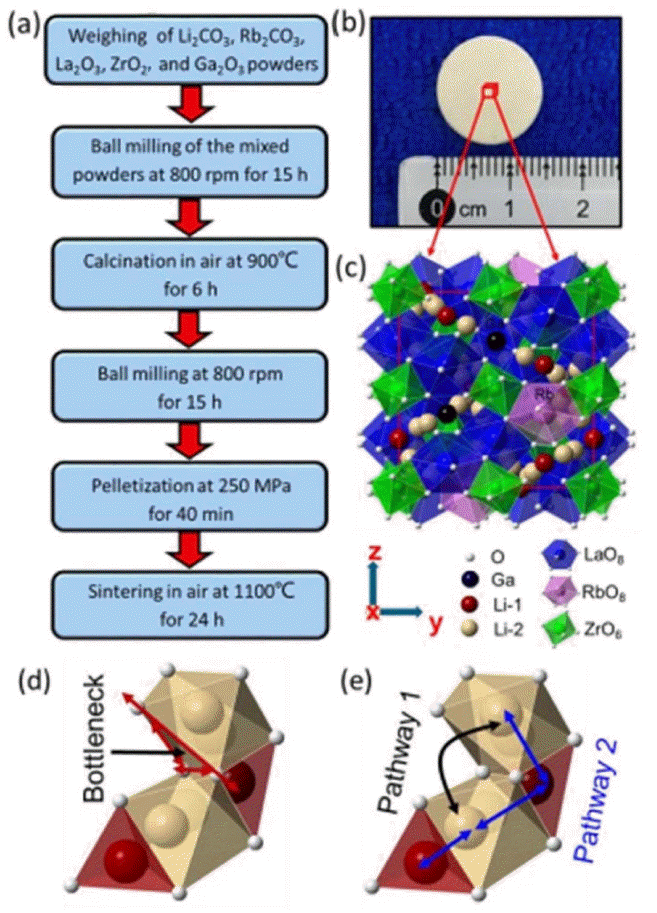
Thus far, LLZO doping modification is mainly concentrated in the Li and Zr sites because the La site has a greater influence on the entire crystal structure of LLZO and determines the bottleneck size of the lithium ion transport. 120) Therefore, it is difficult to find suitable doping ions for the La site. The supervalent doping of the Li and La sites causes lithium vacancies and disordered lithium sublattices. In contrast to supervalent doping, low-valence doping results in excess lithium ions. It was reported that doping at the La site with a relatively large-radius cation can enhance the conductivity of LLZO. Jiang-Fang Wu et al.121) doped cation Rb+ at the La3+ site in cubic Li6.10Ga0.30La3Zr2O12, which led to a highest ionic conductivity of 1.62 mS/cm at 25°C. The Rb+ doping facilitates the densification of the pellet at a lower sintering temperature. Compared with La3+ (0.106 nm), Rb+ has a larger ionic radius of 0.148 nm, which contributes to enlarging the Li+ migration pathway and hence increases the ionic conductivity. The preparation method, crystal structure, and Li+ migration pathway are shown in Fig. 16.
Study122) showed that the substitution of the La site with an alkaline earth metal can effectively increase the lithium content at the Li1 site and reduce the lithium content at the Li2 site in the LLZO structure. The lithium ion at the Li1 site is easier to migrate than at the Li2 site.123,124) Therefore, alkaline earth metals (e.g., Mg, Ca, and Sr) have a positive effect on the improvement of lithium ion conductivity and the reduction of the grain boundary resistance. It seems suitable to replace La with Sr because the ionic radius of Sr2+ is very similar to that of La3+, and Sr can act as a sintering and densification agent.125) The ionic conductivity of the 1.7 wt.% Sr-doped LLZO at room temperature is up to 5.0 × 10−4 S/cm by traditional solid-state synthesis, while the conductivity of the undoped LLZO is only 2.1 × 10−4 S/cm at room temperature.122) In a recent work, a modified solution method with the influence of elements Ca and Ta doped on LLZO was discussed.126,127) The synergetic doping of Ca and Ta was found to induce the formation of cubic LLZO with a high ionic conductivity of 4.03 × 10−4 S/cm.
In addition to the doping of the La site by a monovalent alkali metal and divalent alkaline earth metal, La also can be replaced by a supervalent element such as Ce4+. Ce4+ has a similar ionic radius to La3+ and can appropriately reduce the lithium ion concentration, which is favorable for the improvement of the ionic conductivity.128) Rangasamy et al.128) studied the effects of different doping contents (0 < x < 0.8) on the phase and structure of LLZO by the traditional solid-state sintering method. It was found that when the content of Ce4+ exceeded 0.2, it helped to stabilize the cubic phase lattice structure of LLZO. However, the total ionic conductivity of the Ce-doped LLZO was only 0.014 mS/cm, which was considerably lower than that of the elements substituted by Li and Zr. The lower conductivity was probably caused by CeO2 precipitation at the grain boundaries, which might lead to a high grain boundary resistance.
4.3. Zr site substitution
Compared to LLZO doping modifications in Li and La sites, Zr site doping modification is the most studied method in research on LLZO. It is a commonly used and effective method to stabilize the lattice of cubic-LLZO by Ta49,120,128–135,146,149–151) doping. In addition, there is extensive research interest in this field, including substituting Zr sites with Ge,145) Mo,137) W,138) Sb,139,140) Nb,141,142,152,153) and Y143) ions; Nb/Y co-doping (both doping to Zr sites); Ca/Ta co-doping (Ca doping to Li and La sites, Ta doping to Zr sites); Mg/Ta co-doping (Mg doping to Li sites, Ta doping to Zr sites); and Ba/Ta co-doping (Ba doping to Li and La sites, Ta doping to Zr sites).
Considering ion conductivity, by far, the doping modification method for Zr sites above will effectively increase the ionic conductivity of electrolyte to the magnitude of 10−4 S/ cm. Ta doping can easily achieve 10−3 orders of magnitude among all of these elements. In 2013, Baek128) processed Ta- LLZO with a high ionic conductivity of 1.35 × 10−3 S/cm by spark plasma sintering (SPS). In 2014, Sun et al.132) focused on the density as an impact factor of the ionic conductivity of electrolyte. The researchers fabricated Li6.4La3Zr1.4Ta0.6O12 ceramic disks with a 99.6% density by a hot-press sintering technique. This successfully increased the ionic conductivity of Ta-doped LLZO to a magnitude of 1.6 × 10−3 S/cm.
The method of Zr site doping has the potential to reduce the sintering time and temperature. Compared to the solgel method and SPS, when using the traditional method of solid-state sintering, a higher sintering temperature (> 1100°C) and longer sintering time (> 10 h) are necessarily involved in the process. However, Zr site doping contributes to improving the sintering efficiency, which will shorten the process time of traditional solid-state sintering. In addition, energy conservation has a positive impact. W. Xu et al.136) doped GeO2 of less than 1 wt.% to fabricate Ge-LLZO with an ionic conductivity up to 8.28 × 10−4 S/cm at room temperature. This reduced the sintering time and temperature for the solid-state reaction as well. Yu Tang et al.153) synthesized Nb-LLZO with B2O3 by a solid-state reaction method.
The cubic LLZN phase was obtained after calcining at 850°C for 6 h. However, the ionic conductivity was relatively low compared to other examples: 1.86 × 10−4 S/cm.
In case of a negative impact on the lattice of LLZO, the choice of doping element will be the determinant.29) Not only the performance of the doped LLZO, but also the ability to dope depend on the radius of doping cation. Fig. 17 reveals the relationship between the radius of the doped ion and the highest ion conductivity in Zr sites of LLZO in the research. It can be concluded from the results that the electrochemical performance has been improved when the radius of the doped ion lies in the range of 0.5–0.7 Å. In particular, Te doping and Ta doping will enhance the ion conductivity to 10−3 S/cm.

Summary of dopant ion radiation and ion conductivity of doped LLZO (red circle represents undoped LLZO, purple circles represent doped LLZO).
In the meantime, elements with radii that lie in the typical range, such as Ge4+, Sb5+, and Nb5+, will have a positive impact on ionic conductivity. The ionic conductivity will surpass 6 × 10−4 S/cm after doping these typical elements to Zr sites. It should be noted that all the results above are from the best set of experimental data. Owing to the influence of the sintering process on the material structure and different doping concentrations, the results of the ionic conductivity will turn out to be different even when the same element has been used for doping. For example, the ionic conductivity may still be as low as 2 × 10−4 S/cm146) with single doping, even when doping with the most commonly used element Ta. The disappointing performance is caused by factors such as low density.
Co-doping and single-element doping have attracted extensive research interest. For example, Xiaolan Chen et al.148) co-doped Ca and Ta to LLZO (Ca in the Li and La sites, Ta in the Zr sites). The synergetic effect from Ta and Ca enhanced the ion conductivity to a relatively high magnitude of 2.84 × 10−4 S/cm compared to that of single Ta-doped LLZO (1.95 × 10−4 S/cm). The reason is that Ta plays a key role in stabilizing the cubic phase, and a small amount of Ca compensates the loss of Li content caused by doping a pentavalent Ta ion. However, although co-doping will stabilize the lattice of cubic LLZO, sometimes its ion conductivity may be lower than the single-element doped LLZO of these elements; this aspect needs to be addressed. Xiao Huang et al.156) prepared Ta-LLZO with MgO, which led to a 5.0 × 10−4 S/cm ion conductivity at room temperature. Unfortunately, pure LLZTO has a higher ion conductivity of 7.3 × 10−4 S/cm compared to the result of the above co-doping.
At present, the progress of experimental research on Zr site doping has slowed, while the theoretical calculation is still in progress. At the same time, the study of single doping is basically complete for those elements that have the potential to improve or affect the performance of LLZO electrolyte. While the mechanism of double doping or even multiple- element co-doping is more complicated, research on theoretical analysis and explanation is lacking. This has delayed the experimental study of this topic. As with other sites, Zr site doping can lead to the lattice distortion of the materials, so the size of the lithium ion transmission channel is affected which in turn changes the ionic conductivity of LLZO. In addition to focusing on simple element replacement, it is necessary to study the Zr site doping from more angles and deeper levels, and this is also the future research trend.
5. Conclusions
Garnet-type inorganic solid electrolyte Li7La3Zr2O12 (LLZO) possesses high ionic conductivity (10−4–10−3 S/cm) at room temperature and low electronic conductivity (< 10−11 S/cm), as well as the advantages of good chemical stability against lithium anodes, good thermal stability, and a wide electrochemical window, etc. Thus, it has significant research value and wide practical application prospects in the development of all-solid-state lithium batteries. In recent years, extensive research studies have been conducted on the Liion transport mechanism, element doping, and preparation methods of LLZO in order to improve the ionic conductivity while reducing the manufacturing cost, decreasing the sintering temperature, and shortening the synthesis cycle.
However, to realize the commercialization and practical application of all-solid-state lithium batteries, there are still some issues to be solved. First, LLZO is reactive with atmospheric H2O and CO2 even at room temperature, which deteriorates the ionic conductivity. Thus, the production process is preferably taken under an inert atmosphere, or additives can be used to suppress the unfavorable reaction. Then, the inhibition and mechanism of the lithium dendrite problem need further investigation. Finally, the large interfacial resistance and unstable solid-solid interface contact between LLZO solid electrolyte and electrodes caused by its intrinsic high rigidity are the main problem in its practical application because these strongly affect the cycle stability and rate capability of the batteries. Therefore, the current research hot spot has gradually shifted to interface issues, which are key to the industrial production and practical application of LLZO.
Acknowledgments
This work was financially supported by the National Key Research and Development Program of China (No. 2018YFB0905600 and 2017YFB0310400), the National Natural Science Foundation of China (No. 51472188 and 51521001), Fundamental Research Funds for the Central Universities in China, State Key Laboratory of Advanced Electromagnetic Engineering and Technology (Huazhong University of Science and Technology), and the “111” project (No. B13035).